Estimation of Ammonia Adsorption Capacity on Zeolitic Material Exchanged with Co and Fe Metal Ions
Ⓒ The Korean Environmental Sciences Society. All rights reserved.
This is an Open-Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
In this study, a fusion/hydrothermal method was used to synthesize a zeolite (HDZ) from coal fly ash at an HD thermal power plant. Ammonia adsorption breakthrough experiments and NH₃-TPD measurements revealed that the adsorption performance varied depending on the crystallinity and framework structure of the HDZ. The ammonia adsorption breakthrough curves and capacities were evaluated based on the ion exchange capacities of Fe³⁺ and Co²⁺ incorporated into the HDZ framework, which serves as an adsorbent for ammonia removal. Consequently, the highest ammonia adsorption capacities were observed for metal-ion-supported HDZs, specifically 3 wt% Fe-HDZ (41.35 mg/g) and 4 wt% Co-HDZ (43.38 mg/g), surpassing that of the unmodified HDZ (27.44 mg/g). These enhancements result from the uniform distribution of Fe³⁺ or Co²⁺ ions within the zeolite surface and pores, which increases the Brønsted acid sites and improves ammonia adsorption performance.
Keywords:
Ammonia, Adsorption, Coal fly ash, Zeolitic material, Ion-exchange1. Introduction
Ammonia is emitted from various sources such as industry, agriculture, and municipal waste, and it is recognized as a serious environmental issue in the era of climate change driven by global warming. In the atmosphere, ammonia not only acts as a nitrogen-based odorant that causes an unpleasant smell but also serves as a precursor of secondary fine particulate matter through atmospheric chemical reactions, thereby contributing significantly to air quality deterioration. Despite these environmental hazards, ammonia is widely used in fertilizer production, refrigeration, and the chemical industry. More recently, its importance has been highlighted as a clean energy carrier, making the development of efficient recovery and recycling technologies urgently necessary, beyond simply reducing emissions (Lee et al., 2021). Especially with global efforts to achieve carbon neutrality, the importance of ammonia management has become increasingly critical, and securing effective control technologies is an urgent priority.
Recent research on adsorption for ammonia removal and recovery has shifted from focusing solely on eliminating toxic ammonia to emphasizing high efficiency, selectivity, regenerability, and stability (Bandosz et al., 2009; Ghauri et al., 2012; Rieth et al., 2018). Among various adsorption processes, zeolites (Bernal et al., 1993; Park et al., 2017; Khabzina et al., 2018), activated carbon (Somy et al., 2009; Oktavitri et al., 2017), and metal-organic frameworks (MOFs) are the porous materials most commonly employed as ammonia adsorbents. Specifically, MOFs have been extensively studied due to their precisely tunable pore structures and surface properties, which enable high adsorption capacity and selectivity for ammonia (Rieth et al., 2018; Khabzina et al., 2018). Activated carbon, with its high surface area and porosity, has also been investigated for its adsorption rate, capacity, and mechanism. Although surface modification through acid treatment has been shown to enhance adsorption efficiency, a drawback remains in that functional groups are removed during thermal desorption (Huang et al., 2008; Rieth et al., 2018; Khabzina et al., 2018).
Kim(2016) conducted a study on the removal of ammonia nitrogen using zeolites, focusing on factors such as the crystallographic structure, type of cations, initial concentration, reaction temperature, and solvent conditions. Furthermore, some researchers have reported that the ion-exchange capacity of zeolites and the distribution of adsorbable acidity vary depending on the type and concentration of exchanged cations, thereby influencing the efficiency of ammonia adsorption according to the cationic composition within the zeolite framework (Sherry, 1966; Sherry et al., 1975; Gaus et al., 1981; Drummond et al., 1983; Pebalan et al., 2001).
More recently, studies on ammonia gas removal in the fields of atmospheric science and green hydrogen have been reported, including adsorption studies using zeolite X ion-exchanged with Cu²⁺ and Mg²⁺ (Park et al., 2017), as well as investigations analyzing the adsorption performance of zeolites synthesized from coal fly ash (CFA) and subsequently ion-exchanged (Park et al., 2022; Park et al., 2023).
This study builds upon the work of Park and Lee(2022), who synthesized a zeolite (HDZ), by comparing its ammonia adsorption performance with that of natural, commercial, and reagent-grade zeolites. To enhance the ammonia adsorption performance, the synthesized HDZ was modified into a zeolite-based adsorbent by partially exchanging a cation (Fe³⁺ or Co²⁺). The physicochemical properties of these zeolitic adsorbents were characterized using X-ray diffraction (XRD) and NH₃-TPD analyses, while their ammonia adsorption performance was evaluated through breakthrough curve experiments and adsorption capacity measurements.
2. Materials and Methods
2.1. Preparation of adsorbents
In this study, zeolitic materials used as adsorbents were synthesized following the fusion/hydrothermal synthesis procedure described by Park and Lee(2022). CFA obtained from Hadong Power Station was employed as the raw material. The synthesized zeolite (HDZ: zeolite synthesized using CFA from Hadong Power Station) was repeatedly washed with distilled water by filtration, dried at 105°C for more than 12 h, and subsequently used in the experiments.
To compare the ammonia adsorption performance of the synthesized HDZ, NAZ (natural zeolite), CMZ (commercial zeolite; Cosmo), and WKZ (reagent-grade zeolite; Wako) were also tested using a fixed-bed microreactor.
Furthermore, to enhance the ammonia adsorption capacity, HDZ was stirred in aqueous solutions containing 1–6 wt% of transition metals (Fe or Co) at 30°C for 24 h, followed by three cycles of washing with distilled water through filtration and drying at 105°C for 2 h. In addition, to remove residual chloride ions, the dried zeolites, ion-exchanged with different amounts of transition metals, were calcined at 550°C for 2 h, yielding the final adsorbent samples.
2.2. Experimental apparatus and procedure
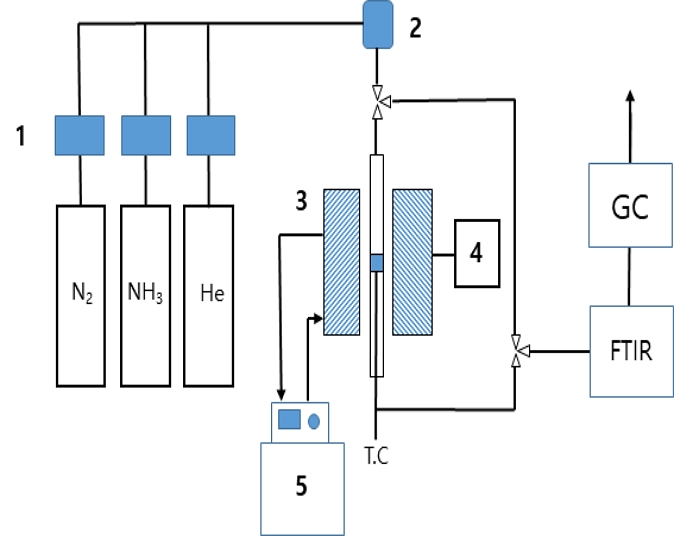
This study was conducted using an atmospheric fixed-bed microreactor; a schematic diagram is shown in Fig. 1. The adsorption reactor was equipped with an IR furnace to control the temperature. Ammonia-adsorbent samples (0.1 g, particle size 120 < dp < 150 ㎛) were packed into a Pyrex reactor tube (outer diameter 6 mm, inner diameter 4 mm) to maintain the isothermal conditions of the reactor. The feed gas (0.1% NH₃/He balance) was supplied at a constant flow rate controlled by a mass flow controller (Brooks). The total flow rate was set to 100 mL/min, and adsorption experiments were performed at ambient temperature. For quantitative analysis of ammonia adsorption, an FT-IR spectrometer (Thermo Scientific™, Nicolet™ Summit PRO FTIR Spectrometer) was used to monitor NH₃ peaks, enabling the determination of breakthrough curves as a function of adsorption time.
2.3. Physicochemical characterization of adsorbents
To investigate the structural changes in the crystalline phase of the synthesized zeolite (Na-A), powder samples were analyzed using an XRD instrument (Rigaku, Primus IV) under the following conditions: 40 kV, 30 mA, and a scan range of 5–50°.
The acidity strength of the ion-exchanged zeolite (HDZ) was examined by ammonia temperature-programmed desorption (NH₃-TPD). For NH₃-TPD analysis, a BETCAT-M instrument (Belcat Inc., Japan) equipped with a thermal conductivity detector (TCD) was employed. The sample weight was 100 mg; the pretreatment conditions are summarized in Table 1. After pretreatment, adsorption was carried out at 30°C using 5 mol/mol% NH₃/He gas at a flow rate of 50 mL/min. The sample was then purged with He, followed by heating to 550°C at a rate of 10°C/min. The desorbed ammonia was monitored using the TCD.
3. Results and Discussion
3.1. Characterization of CFA and zeolite
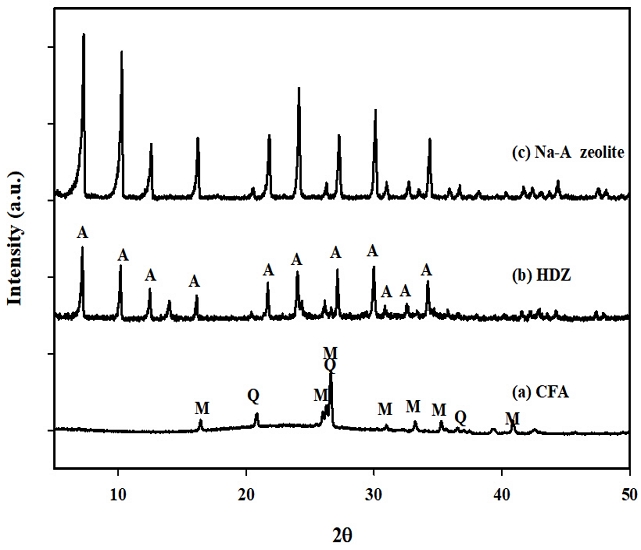
In their study, Park and Lee(2022) selected HDZ, a synthesized zeolite derived from CFA, based on its superior crystallinity and ammonia adsorption breakthrough performance. Fig. 2 shows the XRD patterns of (a) CFA, (b) synthesized zeolite (HDZ), and (c) standard Na-A zeolite. As illustrated in Fig. 2(a), the XRD peaks of CFA were primarily attributed to quartz (Q peak) and mullite (M peak). However, the raw material CFA predominantly exhibited XRD peaks corresponding to quartz (Q peak) and mullite (M peak). However, the HDZ synthesized by mixing NaOH with CFA through the fusion/hydrothermal method was identified as Na-A zeolite, showing characteristic XRD peaks at the same positions as reagent-grade Na-A zeolite (Wako) in Fig. 2(b) and (c).
3.2. Breakthrough curves of ammonia adsorption by different types of zeolites
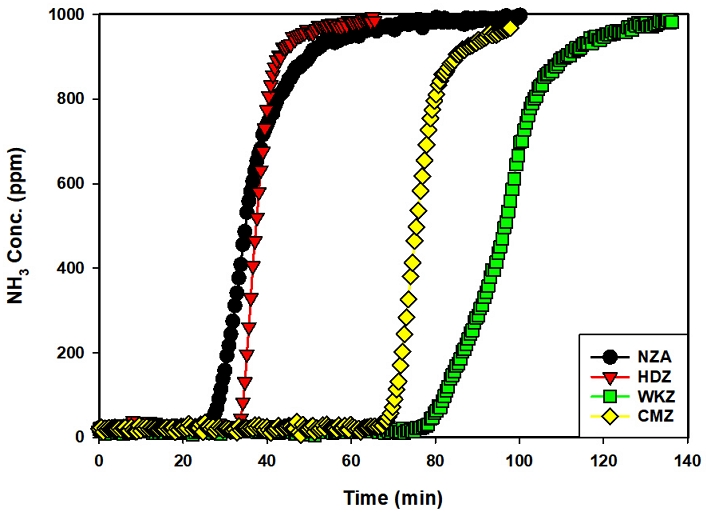
Fig. 3 presents the results of ammonia adsorption breakthrough experiments conducted with four types of zeolites. The breakthrough curves differed depending on the zeolite type, in the following order: WKZ (reagent-grade zeolite) > CMZ (commercial zeolite) > HDZ (synthesized zeolite) > NAZ (natural zeolite). In the case of HDZ, the adsorption capacity for NH₃ gas was superior to that of NAZ; however, it exhibited only about 50% of the adsorption performance of CMZ. The NAZ showed the lowest adsorption efficiency among the tested zeolites, which can be attributed to the presence of impurities, structural defects, and the non-uniform pore distribution (Lee et al., 2019). The synthesized HDZ in this study also contained impurities, resulting in relatively lower crystallinity compared with WKZ and CMZ, which explains the differences observed in adsorption efficiency.
3.3. NH₃-TPD evaluation of adsorbents
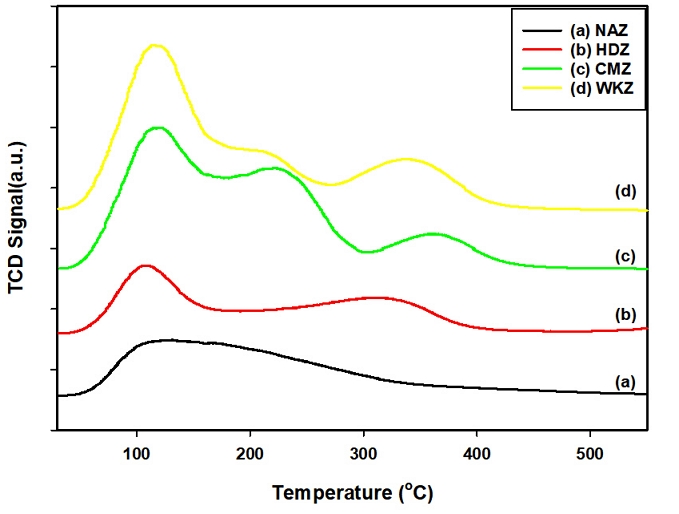
Fig. 4 shows the NH₃-TPD results obtained after investigating the ammonia adsorption characteristics of different types of zeolites. Among (a) NAZ, (b) HDZ, (c) CMZ, and (d) WKZ, (a) NAZ exhibited a single desorption peak, with most ammonia desorbed in the low-temperature (LT) region at around 127°C. In contrast, as shown from (b) to (d) in Fig. 4, multiple desorption peaks were observed, and (b) HDZ displayed an LT desorption peak at 106°C and a high-temperature (HT) desorption peak at 306°C. For (c) CMZ and (d) WKZ, the first LT desorption peak appeared near 118°C, followed by a new middle-temperature (MT) peak at around 212–220°C. The HT desorption peaks shifted to higher temperatures, appearing in the range of 345–360°C. Notably, it shows that the desorption areas of the synthesized zeolites (b–d) were larger than that of (a) NAZ. This indicates that the ammonia adsorption and desorption capacities vary significantly depending on the framework structure and crystallinity of each synthesized zeolite.
Table 2 summarizes the calculated ammonia desorption amounts in the temperature range of 30–550°C in Fig. 4. As shown in the Table 2, WKZ exhibited the highest desorption amount of 2.993 mmol/g, which was approximately three times greater than that of NAZ and HDZ synthesized from CFA. The order of ammonia desorption capacity was WKZ > CMZ > HDZ > NAZ. The relatively low desorption capacity of natural zeolite (NAZ) can be attributed to impurities, non-uniform pore distribution, and poor crystallinity. Although HDZ contained some impurities, its Na-A zeolite framework contributed to a relatively higher desorption capacity compared with NAZ. The enhanced ammonia adsorption performance of the synthesized zeolites is likely due to the more uniform distribution of acidity across the pores and surfaces compared with natural zeolite. Furthermore, during synthesis, lower impurity resulted in more homogeneous pore structures and sizes, which increased the number of alumina sites within the framework. This, in turn, seems to have improved the adsorption performance toward ammonia gas (Benaliouche et al., 2008).
3.4. Breakthrough curves of ammonia adsorption for metal (Fe or Co) ion-exchanged zeolites
The ammonia adsorption characteristics were investigated for HDZ zeolites synthesized from CFA and subsequently ion-exchanged with varying amounts of two transition metals (Fe or Co).
Fig. 5 shows the ammonia adsorption characteristics of the synthesized zeolite HDZ, ion-exchanged with Fe at loadings of 1–5 wt%. The breakthrough curves show that the adsorption performance varied with Fe content, in the following order: 3 wt% > 4 wt% > 5 wt% > 1 wt% > HDZ. Previous studies (Park and Lee, 2022; Park et al., 2023) have reported that when the Fe ion-exchange content exceeds 3 wt%, the incorporation of Fe ions affects the zeolite crystalline framework, thereby reducing the ammonia adsorption capacity.
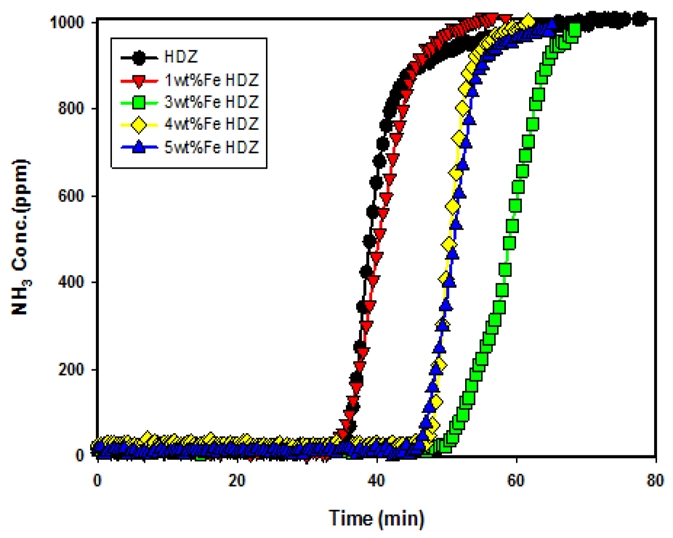
NH₃ adsorption breakthrough curve according to Fe ion exchange content (w= 0.1 g, T= 20℃, Q= 100 mL/min, CA0= 1,000 ppm).
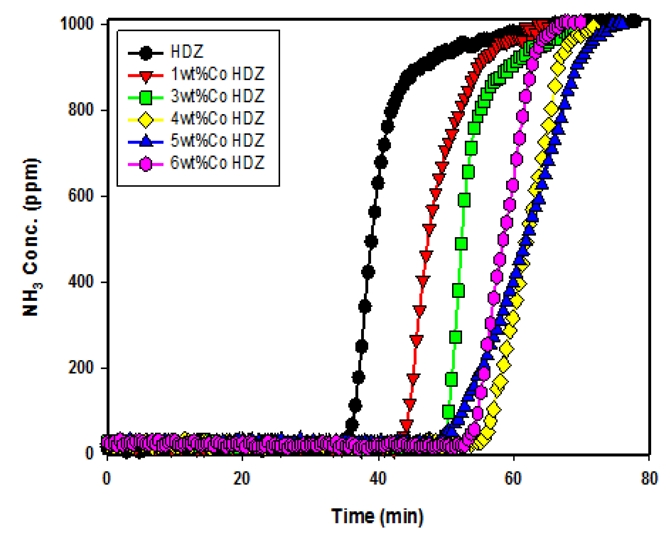
Fig. 6 presents the ammonia adsorption characteristics of the synthesized zeolite HDZ, ion-exchanged with Co at loadings of 1–6 wt%. The breakthrough curves for NH₃ adsorption were observed to be delayed in the order of Co ion-exchange amounts: 4 wt% ≈ 5 wt% > 6 wt% > 3 wt% > 1 wt% > HDZ. It was observed that when the Co ion-exchange content exceeded 4 wt%, the ammonia adsorption capacity decreased. This decline can be attributed to physicochemical changes such as pore blockage or surface distortion caused by higher ion-exchange levels, which reduce the available adsorption capacity (Benaliouche et al., 2008; Kim, 2016).
3.5. Evaluation of ammonia adsorption capacity
The adsorption capacity of the zeolites (mg adsorbate/g adsorbent) was calculated from the breakthrough curves obtained in the fixed-bed adsorption experiments, using the following theoretical equation (Kim et al., 2010; Kim et al., 2012; Park et al., 2022):
| (1) |
where q represents the adsorption capacity (mg adsorbate/g adsorbent); CA0 and CA are the inlet and outlet concentrations of the adsorbate [ppm], respectively; Q is the flow rate of the mixed gas (cc/min); w is the mass of the adsorbent (g); Mw is the molecular weight of the adsorbate; and t is the operating time of the reactor. The breakthrough curves were analyzed using regression analysis with the statistical software SigmaPlot 14.5 to determine CNH3 and t, and the adsorption capacity q was subsequently calculated and is summarized in Table 3.
As shown in Table 3, the adsorption capacities calculated from the breakthrough curves in Fig. 3 were in the following order: WKZ (67.55 mg/g) > CMZ (54.46 mg/g) > HDZ (27.44 mg/g) > NAZ (24.45 mg/g). These results indicate that higher purity and crystallinity of zeolites lead to longer breakthrough times and greater adsorption capacities. Furthermore, the adsorption capacities obtained from the breakthrough curves of 3 wt% Fe-HDZ and 4 wt% Co-HDZ (Fig. 4 and 5) were 41.35 mg/g and 43.48 mg/g, respectively, significantly higher than that of HDZ (27.44 mg/g). This enhancement can be attributed to the ion-exchange of cations within the synthesized zeolite (HDZ) with transition metal ions, which resulted in a more uniform distribution of Brønsted acidity across the pores and surfaces of the zeolite, thereby improving ammonia adsorption performance.
4. Conclusions
Zeolitic material (HDZ) was successfully synthesized from coal fly ash (CFA) using the fusion/hydrothermal method, and its physicochemical properties and ammonia adsorption performance were compared with those of various types of zeolite adsorbents. Further, to enhance the ammonia adsorption performance of HDZ, ion-exchange with two cations (Fe or Co) was conducted, and breakthrough curve and adsorption experiments were performed. The following conclusions were obtained:
1) The ammonia breakthrough time was in the following order: WKZ > CMZ > HDZ > NAZ. The ammonia desorption amounts obtained by NH₃-TPD also showed the same order. Higher purity and crystallinity of zeolites resulted in a more uniform distribution of acidity across the pores and surfaces, thereby extending breakthrough times and improving adsorption/desorption performance.
2) For Fe- and Co-exchanged HDZ, the breakthrough curves appeared in the order of 3 wt% > 4 wt% > 5 wt% > 1 wt% > HDZ (Fe) and 4 wt% ≈ 5 wt% > 6 wt% > 3 wt% > 1 wt% > HDZ (Co). When the ion-exchange content of Fe or Co exceeded the optimal level, the adsorption capacity decreased due to pore blockage or surface distortion caused by structural changes.
3) The adsorption capacities of synthesized and ion-exchanged zeolites, 3 wt% Fe-HDZ and 4 wt% Co-HDZ, were 41.35 mg/g and 43.48 mg/g, respectively, which were higher than that of HDZ (27.44 mg/g). This can be attributed to the ion-exchange of cations within HDZ with transition metal ions, which led to a more uniform distribution of Brønsted acidity across the pores and surfaces of the zeolite, thereby enhancing ammonia adsorption performance.
Acknowledgments
This paper was supported by research fund offered from Catholic University of Pusan in 2022.
REFERENCES
-
Bandosz, T. J., Petit, C., 2009, On the reactive adsorption of ammonia on activated carbons modified by impregnation with inorganic compounds, J. Colloid Interface Sci., 338, 329-345.
[https://doi.org/10.1016/j.jcis.2009.06.039]

-
Benaliouche, F., Boucheffa, Y., Ayrault, P., Mignard, S., Magnoux, P., 2008, NH3-TPD and FTIR spectroscopy of pyridine adsorption studies for characterization of Ag and Cu-Exchanged X Zeolite, Microporous Mesoporous Mater., 111, 80-88.
[https://doi.org/10.1016/j.micromeso.2007.07.006]

-
Bernal, M. P., Lopez-Real, J. M., 1993, Natural zeolites and sepiolite as ammonium and ammonia adsorbent materials, Bioresour. Technol., 43, 27-33.
[https://doi.org/10.1016/0960-8524(93)90078-P]

-
Drummond, D., Jonge, A. D., Rees, L.C. 1983, Ion-Exchange kinetics in Zeolite A, J. Phys. Chem., 87, 1967-1971.
[https://doi.org/10.1021/j100234a027]

-
Gaus, H., Lutze, W., 1981, Kinetics of strontium/barium and strontium/calcium ion exchange in synthetic zeolite A, J. Phys. Chem., 85, 79–84.
[https://doi.org/10.1021/j150601a018]

- Ghauri, M., Tahir, M., Abbas, T., Khurram, M. S., Shehzad, K., 2012, Adsorption studies for the removal of ammonia by thermally activated carbon, Science International, 24, 411-414.
-
Huang, C. C., Li, H. S., Chen, C. H., 2008, Effect of surface acidic oxides of activated carbon on adsorption of ammonia, J. Hazard. Mater., 159, 523-527.
[https://doi.org/10.1016/j.jhazmat.2008.02.051]

-
Khabzina, Y., Farrusseng, D., 2018, Unravelling ammonia adsorption mechanisms of adsorbents in humid conditions, Microporous Mesoporous Mater., 265, 143-148.
[https://doi.org/10.1016/j.micromeso.2018.02.011]

- Kim, S. S., Lee, C. H., Park, S. W., 2010, Adsorption analysis of VOCs of zeolite synthesized from coal fly ash in a fixed-bed adsorber, Korean Chem. Eng. Res., 48, 784-790.
-
Kim, S. S., Lee, C. H., 2012, Adsorption of SO2 by zeolite synthesized from coal fly ash, J. Environ. Sci. Int., 21, 687-694.
[https://doi.org/10.5322/JES.2012.21.6.687]

-
Kim, C. G., 2016, Removal of ammonium and nitrate nitrogen from wastewater using zeolite, J. Korea Org. Resource Recycl. Assoc., 24, 59-63.
[https://doi.org/10.17137/korrae.2016.24.1.59]

-
Lee, K. H., Kil, B. M., Ryu, C. H., Hwang, G. J., 2019, Removal of ammonia nitrogen, manganese and arsenic in the ion exchanged natural zeolite, Membrane Journal, 29, 237-245.
[https://doi.org/10.14579/MEMBRANE_JOURNAL.2019.29.5.237]

-
Lee, H. K., Woo, Y. M., Lee, M. J., 2021, The needs for R&D of ammonia combustion technology for carbon neutrality, J. Korean Soc. Combust., 26, 59-83.
[https://doi.org/10.15231/jksc.2021.26.1.059]

-
Oktavitri, N. I., Purnobasuki, H., Kuncoro, E. P., Purnamasari, I., 2017, Ammonia removal using coconut shell based adsorbent: Effect of carbonization duration and contact time, IPTEK Journal of Proceedings Series, 3, 26-32.
[https://doi.org/10.12962/j23546026.y2017i4.3072]

- Park, J. W., Seo, Y. J., Ryu, S. H., Kim, S. D., 2017, Ammonia adsorption capacity of zeolite X with different cations, Appl. Chem. Eng., 28, 355-359.
-
Park, J. W., Lee, C. H., 2022, Ammonia adsorption capacity and breakthrough curve of zeolitic materials synthesized from coal fly ash, J. Environ. Sci. Int., 31, 833-811.
[https://doi.org/10.5322/JESI.2022.31.10.833]

-
Park, J. W., Kwak, J. Y., Lee, C. H., 2023, Evaluation of ammonia adsorption capacity using various metal ion-exchanged zeolitic materials synthesized from coal fly ash, J. Environ. Sci. Int., 32, 343-353.
[https://doi.org/10.5322/JESI.2023.32.5.343]

-
Pebalan, R. T., Bertetti, F. P., 2001, Cation-exchange properties of nature zeolites, Geochemical Society, 435-518.
[https://doi.org/10.1515/9781501509117-016]

-
Rieth, A. J., Dincă, M., 2018, Controlled gas uptake in metal-organic frameworks with record ammonia sorption, J. Am. Chem. Soc., 140, 3461-3466.
[https://doi.org/10.1021/jacs.8b00313]

-
Sherry, H. W., 1966, The ion-exchange properties of zeolites. I. Univalent ion exchange in synthetic Faujasite, J. Phys. Chem., 70, 1158–1168.
[https://doi.org/10.1021/j100876a031]

-
Sherry, H. S., Walton, H. F., 1975, The ion exchange properties of zeolites, J. Phys. Chem., 71, 1457.
[https://doi.org/10.1021/j100864a042]

-
Somy, A., Mehrnia, M. R., Amrei, H. D., Ghanizadeh, A., Safari, M., 2009, Adsorption of carbon dioxide using impregnated activated carbon promoted by Zinc, Int. J. Greenhouse Gas Control, 3, 249-254.
[https://doi.org/10.1016/j.ijggc.2008.10.003]

Department of Fire and Disaster Prevention, Catholic University of Pusan pjw2131@cup.ac.kr
Department of Environmental Administration, Catholic University of Pusan chlee@cup.ac.kr