
Seed Treatment Effects on Overcoming Morphophysiological Dormancy in Ginseng (Panax ginseng C.A. Meyer)
Ⓒ The Korean Environmental Sciences Society. All rights reserved.
This is an Open-Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
This study evaluated the effects of various seed treatment techniques, including fungicide application, chemical scarification, and osmopriming on germination and early seedling performance of ginseng (Panax ginseng C.A. Meyer). Ginseng seeds exhibited the highest germination percentage at 10°C, which increased in the dark. These results confirm the low-temperature and photoblastic germination characteristics of ginseng seeds. Among the fungicide treatments, 0.25% benomyl-thiram resulted in the highest germination rate. Furthermore, germination was hindered at 20°C or higher for all fungicide treatment concentration. Among the plant growth regulators treated, GA3 50 mg·L-1 resulted in the highest germination percentage. Additionally, although GA3 + kinetin treatment improved germination, this was not observed during early seedling growth. Chemical scarification using various organic and inorganic solvents (acetone, KOH, NaOH) consistently decreased germination and caused seed injury, rendering this approach unsuitable for ginseng seeds. Osmotic priming with PEG (-0.75 MPa) and Ca(NO3)2 significantly enhanced germination performance, with PEG producing the highest final germination without delaying the germination rate compared to the control. These findings indicate that low-temperature incubation, GA3 application, and PEG priming can improve ginseng seed germination performance and support the development of more efficient nursery production systems.
Keywords:
Chemical scarification, Dormancy release, Ginseng germination, Morphophysiological dormancy, Osmopriming1. Introduction
Ginseng (Panax ginseng C.A. Meyer), a perennial medicinal plant of the Araliaceae family, has been cultivated for centuries across Northeast Asia, with Korea as the leading producer. Its 6-10 year production cycle and sensitivity to soil conditions, pests, diseases, and replant failure render ginseng cultivation highly demanding and cost-intensive (Bang et al., 2020).
Traditionally valued for root ginsenosides with tonic, immunomodulatory, and sedative properties, ginseng's commercial importance has expanded with the functional food industry. Notably, shoots exhibit higher phenolic content and antioxidant activity than roots (Oh et al., 2025), while flowers and fruits accumulate substantial ginsenosides (Ma et al, 2021), spurring interest in sprout and vegetative biomass production.
Seed quality critically determines cultivation success, as it governs seedling vigor and field establishment (Suh et al., 2017, 2022). Although seed production commences in year 3, high-germination seeds are reliably obtained only from plants >4 years old (Zhang et al., 2020). Germination rates among cultivars vary widely (47-94%), making seed treatment essential given high seed costs and inconsistent germination (Won et al., 1988).
Fresh ginseng seeds exhibit morphophysiological dormancy: underdeveloped embryos require almost 100 days warm stratification (dehiscence phase) followed by 90 days cold stratification (~4°C) for physiological release (Won et al., 1988; Kwon et al., 2001). This double dormancy severely constrains uniform germination. Numerous approaches target seed coat-imposed barriers, including chemical scarification (Doo et al., 2001; Im et al., 2012), organic solvents (Subbaiah, 1982), hot water, and mechanical scarification (Kang et al., 2000). Results remain inconsistent, varying with treatment intensity and seed physiological status.
Gibberellins (GA) effectively substitute cold stratification by promoting embryo growth and hydrolytic enzyme activity (Kwon et al., 1986a; Bewley and Black, 1994; Lee et al., 2022). Cytokinin co-application further enhances germination (Lee et al., 2018), while novel plant extracts show promise (Jang et al., 2024). However, no comprehensive comparison exists evaluating physical, chemical, and hormonal treatments alongside germination kinetics and seedling vigor in ginseng.
This study systematically assessed PGRs, chemical scarification, osmopriming, and light quality to identify optimal strategies overcoming ginseng's morphophysiological dormancy while maximizing germination uniformity and seedling performance for commercial production.
2. Materials and Methods
2.1. Plant material and assessment of seed morphology
Seeds of a six-year-old landrace of ginseng (Panax ginseng C.A. Meyer) were obtained from the Korea Ginseng Distribution Corporation. Immediately after harvest, pulp was removed and the seeds were washed, surface‑dried, and mixed with sterilized coarse sand at a ratio of 1:3 (v/v). The mixture was placed in aerated plastic containers and stratified at 15 ± 1°C for 90 d, with substrate moisture maintained at 60-70% of field capacity. Containers were ventilated weekly and moisture was adjusted as required.
At the end of warm stratification, seeds were separated into dehisced and non-dehisced groups according to endocarp splitting. Embryo development was examined by longitudinally cutting seeds with a surgical blade and observing embryo and endosperm using a light microscope (Eclipse Ci, Nikon, Japan). Seed length, width and 1000-seed weight were measured with and without seed coats. Embryo and endosperm length and width were recorded for both dehisced and non‑dehisced seeds using a digital Vernier caliper, and weights were determined with an electronic balance (FX-2000i, A&D Company, Korea). All seeds were stored at 5°C in a seed refrigerator until use.
2.2. Seed disinfection and germination response to temperature
To evaluate the influence of fungicide treatment, seeds were immersed in benomyl-thiram wettable powder (Farm Hannong, Korea) at 0.25% or 0.5% (w/v) for 1 h at 15°C. Treated seeds were rinsed thoroughly with distilled water and dried in a laminar-flow hood for 6 h.
Germination tests were conducted in growth chambers set at 10, 15, 20 or 25°C. Fifty seeds were placed on two layers of Whatman No. 2 filter paper in 9cm Petri dishes, with three replicates per treatment. 5 mL of distilled water were added to each dish, and seeds were incubated in darkness. Germination was recorded every 2 d for 40 d.
2.3. Effects of plant growth regulators on germination and seedling vigor
To determine the effects of plant growth regulators (PGRs) on germination and early seedling growth, seeds (10 g per treatment) were immersed in gibberellic acid (GA3) or kinetin solutions for 24 h at 15°C. Solution concentrations for both GA3 and kinetin were 10, 50 and 100 mg·L-1. A combined treatment consisting of GA3 50 mg·L-1 + kinetin 50 mg·L-1 was also included.
Seedling vigor was evaluated using the between-paper (BP) method. Twenty seeds were uniformly placed on BP heavy germination paper, covered with regular germination paper, and incubated at 10°C. After 20 d, hypocotyl length and diameter, root length, fresh weight and dry weight were measured. Dry weight was determined after drying samples at 60°C for 24 h.
2.4. Germination response to organic solvents and seed‑coat scarification
To examine the effects of organic solvents, seeds (10 g) were immersed in 50% or 70% (v/v) methanol, acetone or ethanol solutions. Twenty milliliters of each solution were used, and seeds were incubated at 15°C for 1 h. After treatment, seeds were rinsed thoroughly to remove solvent residues and dried for 6 h in a laminar-flow hood.
Seed-coat scarification treatments included potassium hydroxide (KOH), sodium hydroxide (NaOH) and sulfuric acid (H2SO4). Solutions were prepared at 10% and 30% (v/v), and seeds were treated for 30 min or 1 h. After treatment, seeds were extensively rinsed to remove residual chemicals and dried for 6 h.
For all treatments, fifty seeds were placed in Petri dishes with two layers of Whatman No. 2 filter paper, with three replications. Germination was conducted at 10°C in darkness.
2.5. Seed priming treatments
Priming treatments consisted of KCl, KNO3, Ca(NO3)2 and polyethylene glycol (PEG 8000). The PEG solution was adjusted to a water potential of -0.75 MPa following Michel(1983). KCl, KNO₃, and Ca(NO3)2 solutions were prepared at 200 mM. For each treatment, 150 seeds were immersed in 20 mL of priming solution in 50 mL containers and incubated at 15°C for 5 d. After priming, seeds were rinsed, dried for 6 h, and germinated at 10°C for 40 d with germination assessed daily.
2.6. Effects of light quality on seed germination
To evaluate germination responses to light quality, seeds were exposed to red light (632 nm), blue light (460 nm), mixed red:blue light (8:2) or darkness (control). LED light sources were positioned 30 cm above the seed surface. All lighting treatments were applied continuously (24 h·d-1), and germination temperature was maintained at 10°C.
2.7. Germination criteria and statistical analysis
Seeds were considered germinated when the radicle reached at least 1 mm in length. Germination was recorded every 1-2 d for a total of 40-60 d, depending on the experiment. Time to 50% germination (T50) was calculated following Coolbear et al. (1984). Variables in the T50 equation were defined as:
N: Total number of seeds germinated by the end of the test
Nᵢ: Cumulative germination number before reaching 50% of N
Nⱼ: Cumulative germination number just after exceeding 50% of N
Tᵢ: Time corresponding to Nᵢ
Tⱼ: Time corresponding to Nⱼ
Data were analyzed using SAS version 9.4 (SAS Institute Inc., Cary, NC, USA). Analysis of variance (ANOVA) was performed to determine significant treatment effects, followed by Duncan's multiple range test (DMRT) at P < 0.05.
3. Results and Discussion
3.1. Embryo development following stratification
Ginseng (Panax ginseng C. A. Meyer) seeds are generally spherical to ovoid and possess a hard, yellowish seed coat characteristic of hard seeds. Although the seeds appear morphologically mature at harvest, the embryo remains underdeveloped, a condition classified as morphological dormancy (Won et al., 1988). In seeds exhibiting morphological dormancy, the degree of embryo development is a critical determinant of germination onset (Kwon et al., 1986b; Baskin and Baskin, 2004).
Following stratification, embryos of dehisced seeds developed markedly during the 3-month warm phase, accompanied by natural splitting of the seed coat. The embryo and endosperm structures became clearly visible, indicating substantial progress in embryonic maturation (Fig. 1; Table 1). In contrast, embryos were barely discernible in non-dehisced seeds, suggesting that morphological dormancy was largely maintained. These observations confirm that stratification promotes embryo growth and establishes the physiological and morphological conditions necessary for germination.
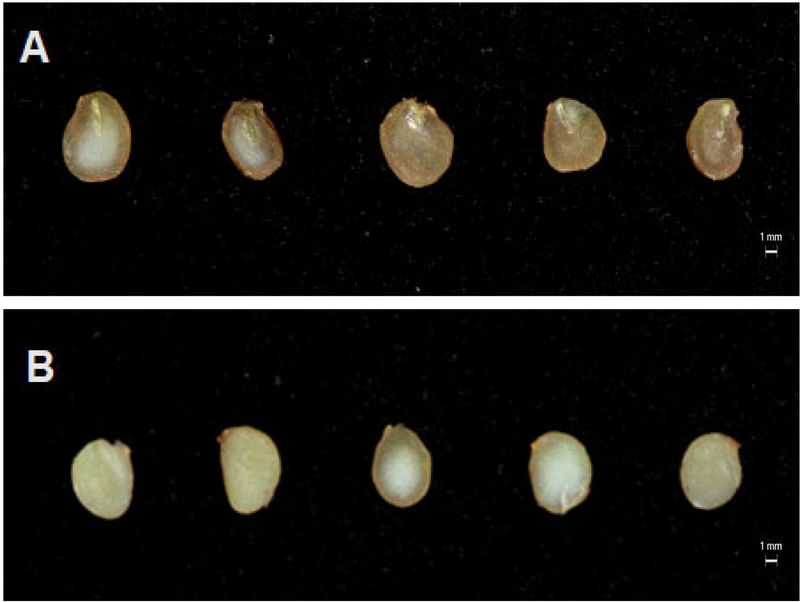
Morphological changes in the embryo and endosperm of ginseng (Panax ginseng C.A. Meyer) seeds following stratification. (A) Non-stratified seeds showing rudimentary embryo. (B) Stratified, dehisced seeds with elongated embryo and loosened endosperm. Scale bar = 1 mm.
Effects of stratification on seed weight and morphological characteristics of ginseng (Panax ginseng C.A. Meyer) seeds
Seed dimensions (length and width) did not differ significantly between dehisced and non-dehisced seeds (Table 1). However, 1,000-seeds weight tended to increase in dehisced seeds, implying enhanced dry matter accumulation associated with increased metabolic activity and water absorption during embryo development (Suh et al., 2022). Specifically, dehisced embryos attained 0.82 mm length and 2.14 mm width, whereas non-dehisced embryos were undetectable.
These results align with reports that embryo length ratio, rather than endocarp dehiscence per se, more reliably predicts germination potential(Kwon et al., 2001; Zhang et al., 2020).
3.2. Effects of fungicide treatment and temperature on germination traits
Fungicide treatment suppresses seed-borne pathogens to promote germination uniformity and seedling establishment (Park et al., 2021). Here, final germination percentage and T50 were assessed across benomyl–thiram concentrations (0.25, 0.5%) and temperatures (10-25°C) (Table 2). Germination occurred exclusively at 10°C and 15°C; no germination was observed at ≥20°C regardless of fungicide concentration. These results confirm ginseng's absolute low-temperature requirement for germination (Kwon et al., 2001).

Effects of germination temperature and benomyl-thiram concentration on germination percentage and T50 in ginseng (Panax ginseng C.A. Meyer) seeds
At 10°C, 0.25% benomyl–thiram resulted in the highest final germination (22.0%), while germination declined at 15°C across all concentrations. Although 0.5% benomyl-thiram also supported substantial germination at 10°C, higher concentrations conferred no additional benefit. Unlike plasma treatments that simultaneously sterilize and modify seed coat permeability (Park et al., 2021), benomyl-thiram primarily controls pathogens without structurally altering dormancy barriers in ginseng.
T50 at 10°C was shortest with 0.5% benomyl-thiram (10.8 days), suggesting pathogen suppression accelerates germination onset. However, T50 increased markedly at 15°C across treatments, indicating temperature exerts primary control over germination kinetics.
Thus, benomyl–thiram enhanced germination stability primarily through pathogen control, while low temperature (10°C) remained the dominant determinant of germination success (Won et al., 1988, Im et al., 2012).
3.3. Effects of plant growth regulators on ginseng seed germination
The effects of plant growth regulator (PGR) type and concentration on ginseng seed germination at 10°C are summarized in Table 3. ANOVA indicated significant main effects of PGR type and treatment concentration(P < 0.001), and their interaction (P < 0.01) on final germination percentage, and highly significant effects on T50 (P < 0.001).

Effects of plant growth regulators and their concentrations on germination percentage and T50 of ginseng (Panax ginseng C.A. Meyer) seeds at 10°C
Across concentrations, GA3 consistently yielded higher germination than the control or kinetin treatments. Maximum germination (47.3%) occurred at 50 mg·L-1 GA3, while germination declined slightly at 100 mg·L-1 GA₃ (43.3%). Gibberellins promote germination through enhanced hydrolytic enzyme activity and embryo elongation (Bewley et al., 2012), consistent with GA3's superior performance in overcoming ginseng's deep morphophysiological dormancy. The lack of dosage response above 50 mg·L-1 suggests an optimal concentration threshold.
Kinetin improved germination over control but remained inferior to GA3, aligning with cytokinins' primary role in cell division rather than dormancy release (Kwon et al., 1986a; Kucera et al., 2005). T50 values followed the same hierarchy, with GA₃ treatments exhibiting the shortest germination time (14.3-14.9 days), followed by kinetin (14.7–16.8 days), and control showing the longest (17.1 days).
These findings demonstrate that GA3 at ≥50 mg·L-1 effectively substitutes for extended stratification by directly targeting morphological and physiological dormancy barriers, while cytokinins provide supplementary benefits (Taiz et al., 2015).
Although germination in the treated groups improved compared with the untreated group, where the final germination rate remained at 47%, which is lower than the typical germination level reported for well-dehisced, high-quality ginseng seeds(Lee et al., 2018). This outcome is likely attributable to variability in seed quality and heterogeneous dormancy depth associated with landrace seed lots, including differences in harvest timing, seed maturity, embryo development, and storage history. Therefore, despite the effectiveness of low-temperature, dark conditions, GA3, and PEG treatments in promoting dormancy release, intrinsic limitations in seed viability likely constrained the overall germination rate.
3.4. Interactive effects of GA3 and kinetin on germination and seedling vigor
The germination responses to GA3 alone and in combination with kinetin are presented in Table 4. GA3 at 50 mg·L-1 significantly increased germination (42.3%) compared with the untreated control. The combined treatment of GA3 50 mg·L-1 + kinetin 50 mg·L-1 produced the highest germination percentage (46.7%), confirming synergistic effects between the two regulators (Bewley and Black, 1994).
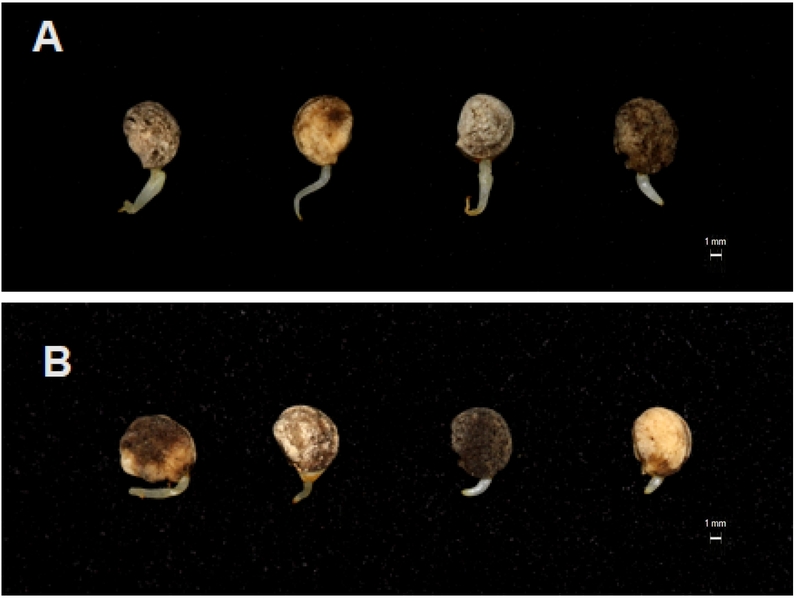
Appearance of ginseng seedlings germinated under plant growth regulator treatments at 10°C. (A) Seeds treated with GA₃ (50 mg·L-1) + kinetin (50 mg·L-1); (B) untreated.

Effects of combined GA3 and kinetin seed treatments on germination percentage and time to 50% of final germination (T50) in ginseng seeds at 10°C
Similarly, T50 was reduced from 17.4 days in the control to 14.9 days in the GA3 50 mg·L-1 treatment and to 13.5 days in the combined treatment. Combined treatment accelerated germination onset while maintaining uniformity at 10°C
However, early seedling growth (Table 5) was greatest in the GA3 50 mg·L-1 single treatment, which exhibited the longest hypocotyls, enhanced root development, and the highest fresh and dry biomass. In contrast, the GA3 + kinetin treatment, although superior for germination initiation, produced less vigorous seedlings. This suggests Kinetin antagonized GA3-mediated cell elongation during seedling establishment (Arteca, 1996).

Effects of seed treatments with gibberellic acid (GA3) alone or in combination with kinetin on early seedling growth and biomass accumulation of ginseng evaluated 20 days after germination at 10°C
Overall, the combined GA3 + kinetin treatment (50 mg·L-1 each) was most effective for germination promotion, whereas GA3 50 mg·L-1 alone was more suitable for enhancing early seedling vigor. These results indicate that the optimal growth regulator treatment may differ depending on whether the production objective emphasizes germination uniformity or seedling quality.
3.5. Germination responses to organic solvent treatments
Organic solvent treatments have been used in some hard-seeded species to increase seed coat permeability and facilitate germination (Khan et al., 1973). However, all treatments failed to promote ginseng seed germination and frequently damaged embryos (Table 6). Trace germination (0.7%) occurred only with 50% methanol and ethanol; acetone treatments yielded 0% germination regardless of concentration or duration. These results confirm that chemical scarification cannot overcome ginseng's morphophysiological dormancy (Won et al., 1988). Unlike simple physical dormancy, ginseng requires coordinated embryo growth and physiological activation, which solvents disrupt through lipid solubilization, membrane damage, and ROS accumulation (Finch-Savage and Leubner-Metzger, 2006; Bewley et al., 2012).

Effects of organic solvent type, concentration, and immersion duration on germination percentage and time to 50% of final germination (T50) of ginseng seeds at 10°C
Thus, organic solvent scarification is unsuitable for ginseng due to embryo toxicity and germination failure.
3.6. Effects of chemical scarification on germination
Chemical scarification using KOH, NaOH, or H2SO4 is commonly employed to break physical dormancy in impermeable seeds. However, all treatments inhibited ginseng seed germination (Table 7). Germination declined markedly at 10% KOH, which further dropped to below 0.7% at 30% KOH. Reduced T50 in some treatments likely reflects accelerated embryo deterioration rather than enhanced germination potential (Gao et al., 1998; Kim et al., 2013). Seed coat penetration increased water uptake but embryo damage predominated.

Effects of chemical stratification agent, concentration, and immersion duration on germination percentage and time to 50% of final germination (T50) of ginseng seeds at 10°C
Similarly, NaOH treatments resulted in severe germination inhibition across all concentrations. Sulfuric acid treatments also strongly suppressed germination, indicating that the embryo is highly sensitive to corrosive chemical scarification.
Collectively, these findings demonstrate that chemical scarification is inappropriate for breaking dormancy in ginseng seeds and may substantially impair seed viability. Further studies exploring narrower concentration ranges or short-exposure protocols may help clarify whether any safe and effective application exists; however, based on the present results, chemical scarification cannot be recommended for ginseng seed pretreatment.
3.7. Effects of priming treatments on germination
Priming responses varied markedly by agent (Table 8). PEG osmopriming (-0.75 MPa) substantially improved germination: final percentage increased from 26.0% (Untreated) to 32.3%, with T50 reduced from 17.9 to 17.3 days. This confirms osmopriming advances metabolic competence without radicle protrusion under low-temperature conditions (Bewley et al., 2012).

Effect of seed priming on germination percentage and time to 50% of final germination (T₅₀) of ginseng seeds germinated at 10°C
Calcium nitrate [Ca(NO3)2] priming also resulted in relatively high germination percentage, though not as high as in PEG. In contrast, KNO3 priming produced the lowest germination and longest T50 among all treatments, indicating nitrate-induced osmotic/ionic toxicity detrimental to ginseng embryo viability. Similar observations have been reported in other species, where excessive solute concentrations during priming can reduce seed vigor (Quintero et al., 2018).
Overall, PEG -0.75 MPa was the most effective priming treatment for enhancing germination capacity under low-temperature conditions in ginseng seeds.
3.8. Effects of light quality on germination
Light quality strongly influenced germination, confirming ginseng's negative photoblasty (Table 9). Seeds germinated most readily and rapidly under dark conditions, whereas red LED and red-blue mixed light produced intermediate germination levels. Blue LED light resulted in the lowest germination percentage and the longest T50 (27.4 d).
These responses align with the ecological germination niche of ginseng, which naturally germinates under leaf litter where low light and cool temperatures prevail (Proctor and Bailey, 1987; Cho et al., 2008). The inhibitory effect of blue light implicates cryptochrome signaling, which upregulates ABA biosynthesis and dormancy maintenance pathways (Folta and Childers, 2008).
Collectively, these findings demonstrate that low temperature (10°C) combined with dark conditions provides the optimal environment for stable and uniform germination of ginseng seeds.
4. Conclusions
This study demonstrates that low-temperature germination (10°C), darkness, GA3 application (50 mg·L-1), and PEG priming (-0.75 MPa) most effectively overcome ginseng's morphophysiological dormancy. Benomyl–thiram (0.25%) provided supplementary pathogen control without compromising germination potential. Chemical scarification (organic solvents, KOH, NaOH, H2SO4) proved uniformly ineffective due to embryo toxicity, confirming that seed-coat permeability is not the primary dormancy barrier in ginseng. Combined GA3 + kinetin maximized germination capacity but reduced early seedling vigor compared to GA3 alone. These optimized protocols enhance germination uniformity and seedling establishment, offering practical solutions for commercial ginseng nursery production while minimizing chemical inputs.
Acknowledgments
This work was supported by a 2-Year research grant of Pusan National University.
REFERENCES
-
Arteca, R. N., 1996, Plant growth substances: Principles and spplications, Chapman & Hall, New York, USA.
[https://doi.org/10.1007/978-1-4757-2451-6]

-
Bang, K. H., Kim, Y. C., Lee, J. W., Cho, L. H., Hong, C. E., Hyun, D. Y., Kim, J. U., 2020, Major achievement and prospect of ginseng breeding in Korea, J. Kor. Breed. Sci., 52, 170-178.
[https://doi.org/10.9787/KJBS.2020.52.S.170]

-
Baskin, J. M., Baskin, C. C., 2004, A Classification system for seed dormancy, Seed Sci. Res., 14, 1-16.
[https://doi.org/10.1079/SSR2003150]

-
Bewley, J. D., Black, M., 1994, Seeds: Physiology of development and germination, 2nd ed., Plenum Press, New York, USA.
[https://doi.org/10.1007/978-1-4899-1002-8]

-
Bewley, J. D., Bradford, K. J., Hilhorst, H. W., Nonogaki, H., 2012, Seeds: Physiology of development, germination and dormancy, 3rd ed., Springer Science Business Media, New York, USA.
[https://doi.org/10.1007/978-1-4614-4693-4]

- Cho, J. S., Lee, L. G., Park, C. H., 2008, Effects of light quality on germination and seedling growth of Panax ginseng, Kor. J. Med. Crop Sci., 16, 385-390.
-
Coolbear, P., Francis, A., Grierson, D., 1984, The effect of low temperature pre-sowing treatment on the germination performance and membrane integrity of artificially aged tomato seeds, J. Exp. Bot., 35, 1609-1617.
[https://doi.org/10.1093/jxb/35.11.1609]

- Doo, H. S., Baek, W. J., Ryu, J. H., 2001, Effects of scarification and soaking treatment on germination of sword bean seed, J. Kor. Crop Sci., 46, 165-169.
-
Finch-Savage, W. E., Leubner-Metzger, G., 2006, Seed dormancy and the control of germination, New Phytologist., 171, 501-523.
[https://doi.org/10.1111/j.1469-8137.2006.01787.x]

-
Folta, K. M., Childers, K. S., 2008, Light as a growth regulator: Controlling plant biology with narrow-bandwidth illumination, HortScience., 43, 1957-1964.
[https://doi.org/10.21273/HORTSCI.43.7.1957]

-
Gao, Y. P., Zhang, G. H., Gusta, L. V., 1998, Potassium hydroxide improves seed germination and emergence in five native plant species, Hort Science., 33, 274-276.
[https://doi.org/10.21273/HORTSCI.33.2.0274]

-
Im, M. H., Kim, B. W., Park, Y. S., Yang, S. Y., Song, C. E., Heo, B. G., 2012, Effects of scarification, temperature and sulfuric acid treatments on seed germination of white lotus (Nelumbo nucifera), J. Kor. Plant Res., 25, 7-13.
[https://doi.org/10.7732/kjpr.2012.25.1.007]

- Jang, M. H., Park, H. W., Mun, G. I., Lee, J. H., Kwon, Y. S., 2024, Effect of natural extract soaking treatment on the dehiscence and germination of ginseng (Panax ginseng C.A. Meyer) seeds, J. Kor. Soc. Med. Crop Sci., 32, 45-53.
- Kang, J. S., Choi, Y. W., Son, B. G., Ahn, C. K., Cho, J. L., 2000, Effect of hydropriming to enhance the germination in gourd seeds, J. Kor. Soc. Hort. Sci., 41(6), 559-564.
-
Khan, A. A., Tao, K. L., Roe, C. H., 1973, Application of chemicals in organic solvents to dry seeds, Plant Physiology, 52, 79-81.
[https://doi.org/10.1104/pp.52.1.79]

-
Kim, M. T., Lee, T. H., Jeon, W. T., Kim, S. J., Yun, D. H., Ku, J. H., Song, H. N., Lee, H. B., Seo, M. C., Kang, H. W., 2013, Effects of water-soaking and mechanical and chemical scarifications on seed germination of hairy vetch (Vicia villosa Roth), J. Kor. Soil Sci. Fert., 46(1), 49-52.
[https://doi.org/10.7745/KJSSF.2013.46.1.049]

-
Kucera, B., Cohn, M. A., Leubner-Metzger, G., 2005, Plant hormone interactions during seed dormancy release and germination, Seed Sci. Res., 15, 281-307.
[https://doi.org/10.1079/SSR2005218]

- Kwon, W. S., Jung, C. M., Ahn, S. D., Choi, K. T., 1986a, Effects of growth regulators on the germination of Panax ginseng C.A. Meyer, J. Kor. Ginseng Sci., 10, 159-166.
- Kwon, W. S., Lee, J. H., Lee, M. K., 2001, Optimum chilling terms for germination of the dehisced ginseng (Panax ginseng C.A. Meyer) seed, J. Kor. Ginseng Sci., 25, 167-170.
- Kwon, W. S., Lee, M. G., Chung, C. M., 1986b, Effects of temperature and stratification on germination of dehisced ginseng seeds, Kor. J. Crop Science., 31, 220-226.
-
Lee, J. W., Jo, I. H., Kim, J. U., Hong, C. E., Kim, Y. C., Kim, D. H., Park, Y. D., 2018, Improvement of seed dehiscence and germination in ginseng by stratification, gibberellin, and/or kinetin treatments, Horticulture, Environment, and Biotechnology, 59, 293–301.
[https://doi.org/10.1007/s13580-018-0039-6]

- Lee, S. S., Cho, K. Y., Won, J. Y., 2018, Improvement of seed dehiscence and germination in ginseng by stratification, gibberellin, and kinetin treatments, Plant Biotechnol. Rep., 12, 245-252.
-
Lee, S. Y., Park, K. T., Jang, B. K., Ji, B. R., Lee, H. M., Baskin, C. C., Cho, J. S., 2022, Exogenous gibberellin can effectively and rapidly break intermediate physiological dormancy of Amsonia elliptica seeds, Front. Plant Sci., 13, 1043897
[https://doi.org/10.3389/fpls.2022.1043897]

-
Ma, L. J., Ma, N., Wang, B. Y., He, X., Wan, J. B., 2021, Ginsenoside distribution in different architectural components of Panax notoginseng inflorescence and infructescence, J. of Pharm, Biomed. Anal., 203, 114221
[https://doi.org/10.1016/j.jpba.2021.114221]

-
Michel, B. E., 1983, Evaluation of the water potentials of solutions of polyethylene glycol 8000 both in the absence and presence of other solutes, Plant Physiology, 72, 66–70.
[https://doi.org/10.1104/pp.72.1.66]

-
Oh, G., Lee, D. S., Chae, J. W., Park, S. W., Im, J. H., Fu, X., Lim, J. S., Kim, M. H., Seong, Y. S., Wei, S., Fu, X., La, I. J., Lee, O. H., 2025, Assessment of 12 ginsenosides and the antioxidant activity of red ginseng sprout extracts, Appl. Sci., 15(23), 12467.
[https://doi.org/10.3390/app152312467]

-
Park, S. C., Kim, J. H., Lee, J. E., Kim, H. S., 2021, Enhancement of seed germination and microbial disinfection on ginseng by cold atmospheric plasma treatment, J. Ginseng Res., 45, 582-589.
[https://doi.org/10.1016/j.jgr.2020.12.002]

-
Proctor, J. T., Bailey, W. G., 1987, Ginseng: Industry, botany, and culture, Horticultural Reviews, 9, 187-236.
[https://doi.org/10.1002/9781118060827.ch6]

-
Quintero, C. M. F., Castillo, O. G., Sánchez, P. D., Marín-Sánchez, J., Guzmán, A. I., Sánchez, A., Guzmán, J. M., 2018, Relieving dormancy and improving germination of piquín chili pepper (Capsicum annuum var. glabriusculum) by priming techniques, Cogent Food & Agric., 4(1), 1550275.
[https://doi.org/10.1080/23311932.2018.1550275]

-
Suh, S. J., Jang, I. B., Yu, J., Jang, I. B., Park, H. W., Seo, T. C., Kweon, K. B., 2017, Effect of seed dehydration and temperature during cold stratification on the seed quality of Panax ginseng C.A. Meyer, J. Kor. Medi. Crop Sci., 25, 209-216.
[https://doi.org/10.7783/KJMCS.2017.25.4.209]

- Suh, S. J., Yu, J., Jang, I. B., Kim, Y. C., 2022, Effect of seed moisture content on seed storage of dehisced ginseng seeds, Kor. J. of Plant Res., 35, 183-191.
-
Subbaiah, C. C., 1982, Effect of pre-soaking in organic solvents on seed germination and seedling growth of cashew, Sci. Hort., 18(2), 137-142.
[https://doi.org/10.1016/0304-4238(82)90128-5]

- Taiz, L., Zeiger, E., Møller, I. M., Murphy, A., 2015, Plant physiology and development, 6th ed., Sinauer Associates, Sunderland, MA, USA.
- Won, J. Y., Jo, J. S., Kim, H. H., 1988, Studies on the germination of Korean ginseng (Panax ginseng C.A. Meyer) seed, II: Influences of temperature and seed treatment on embryo growth and germination, J. Kor. Crop Sci., 33(1), 59-53.
-
Zhang, H., Abid, S., Ahn, J. C., Mathiyalagan, R., Kim, Y. J., Yang, D. C., Wang, Y., 2020, Characteristics of Panax ginseng cultivars in Korea and China, Molecules., 25(11), 2635.
[https://doi.org/10.3390/molecules25112635]

Department of Horticultural Bioscience, Pusan National Universityogihan@korea.kr
Department of Horticultural Bioscience, Pusan National Universitypark5520@pusan.ac.kr
Department of Horticultural Bioscience, Pusan National Universitybije@pusan.ac.kr
Department of Horticultural Bioscience, Pusan National Universitykangjs@pusan.ac.kr