
Effects of Seedling-Stage Diniconazole Application on Tomato Transplant Quality, Early Growth, and Initial Yield
Ⓒ The Korean Environmental Sciences Society. All rights reserved.
This is an Open-Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
This study investigated the carry-over effects of diniconazole, applied during the tomato nursery stage, on seedling quality, early post-transplantation growth, and initial yield under protected cultivation conditions. Diniconazole decreased plant height and internode elongation in a concentration-dependent manner. The 125 mg/L treatment produced the most vigorous seedlings, exhibiting balanced shoot-root development, sustained chlorophyll content, and enhanced node differentiation, whereas 250 mg/L caused excessive growth inhibition and reduced biomass. The carry-over effects persisted throughout early greenhouse cultivation. Plants treated with 125 mg/L showed improved early fruit set, advanced harvest initiation, and a significantly higher cumulative early yield than the control plants. Although the 250 mg/L treatment promoted floral initiation, fruit enlargement was restricted, which reduced total yield. These results identified 125 mg/L diniconazole as the optimal application rate for producing compact, robust seedlings that enhance early season productivity in tomatoes.
Keywords:
Diniconazole, Growth regulation, Seedling quality, Tomato, Triazole1. Introduction
Tomato (Solanum lycopersicum L.) is a major vegetable crop grown widely in controlled-environment greenhouses, where seedling quality strongly influences post-transplant establishment, early reproductive development, and yield potential. Poor-quality or elongated seedlings are prone to weak stems, reduced stress tolerance, delayed inflorescence development, and lower early yields (Kubota et al., 2008; Schwarz et al., 2010). Etiolation typically arises under suboptimal nursery conditions, such as high temperature, low light, and dense spacing, leading to excessive internode elongation and reduced structural stability (Myster and Moe, 1995). Therefore, producing compact, physiologically vigorous seedlings is essential for stable long-term productivity in extended tomato cropping systems (Heins et al., 1998; Fan et al., 2013).
Plant growth regulators (PGRs), particularly triazole compounds, are commonly used during the nursery stage to inhibit gibberellin (GA) biosynthesis and suppress shoot elongation. Diniconazole is an effective GA-inhibiting triazole that produces compact seedlings with reduced internode length (Fletcher et al., 2000; Jang et al., 2020). However, its effects depend strongly on concentration, as excessive application can limit biomass accumulation and negatively affect post-transplant performance (Rademacher, 2000). This creates a critical trade-off for growers: while diniconazole effectively prevents etiolation in the nursery, its residual inhibitory effects may persist after transplanting, potentially delaying the transition to the reproductive stage. Since early-season productivity is vital for economic profitability in greenhouse tomato production, thus, it is imperative to verify that chemical growth regulation does not compromise the plants' ability to recover and initiate flowering promptly. Although related triazoles such as uniconazole have been shown to influence early inflorescence development and initial yield (Dunn et al., 2022), comprehensive studies examining the specific carry-over effects of diniconazole on the continuity between vegetative recovery and early reproductive success under practical greenhouse conditions remain limited (Blanchard and Runkle, 2007; Ali et al., 2022).
This study aimed to evaluate (1) morphological and physiological responses of tomato seedlings to varying diniconazole concentrations during the nursery stage, and (2) the carry-over effects on post-transplant growth, reproductive development, and early yield. The ultimate objective was to determine an optimal level of diniconazole application dosage that enhances seedling quality and supports stable year-round production in protected tomato cultivation.
2. Materials and Methods
2.1. Planting material and seedling growth parameters at nursery stage
A nursery stage experiment was conducted to evaluate the effects of diniconazole on tomato seedling growth. Seeds of the European-type cultivar ‘Redkhan’ (Solanum lycopersicum L.; Mifuko Seed Co., Korea) were sown in 32-cell plug trays filled with a commercial cocopeat–peatmoss–perlite–zeolite substrate (Field King; Nongwoobio Co., Korea). Seedlings were grown for 7 weeks (10 April to 30 May 2025) in a controlled-environment growth chamber (EH-3600, Hi-point, Taiwan) at 25°C, 70% relative humidity, and ambient CO₂ (≈420 μmol/mol). A multi-wavelength LED system supplied 300 μmol·m-2·s-1 PPFD under a 16h photoperiod. Subirrigation was applied when substrate moisture reached approximately 60% of container capacity inorder to avoid any water deficit conditions.
Diniconazole (5% SC; Dongbang Agro Co., Korea) was applied as a foliar spray at 0, 50, 125, or 250 mg/L at 3, 4, and 5 weeks after sowing (2–6 true-leaf stage). Applications were made to runoff, and all cultural conditions were kept uniform among treatments. The experiment followed a completely randomized design with three replications.
For growth measurements and statistical analysis, nine uniform plants per treatment (n = 9) were used as the experimental units. Parameters included plant height, stem diameter, leaf and node number, leaf blade dimensions, and shoot and root biomass. Leaf area was determined using a leaf area meter (LI-3100C, LI-COR, USA). Fresh and dry weights were recorded after drying tissues at 70°C for 72 h. Chlorophyll content was measured on the third fully expanded leaf using a SPAD-502 meter (Minolta, Japan)
2.2. Carry-over effects on early growth and yield after transplanting
Seedlings obtained from the nursery stage were transplanted on 30 May 2025 into a plastic greenhouse in Haman, Gyeongsangnam-do, and cultivated for 13 weeks. Plants were grown on coir slabs (chip:dust = 5:5; Shinhan Atec, Korea) at a density of 3.125 plants/m2 under a randomized complete block design with three replicates. Each treatment consisted of 21 plants (7 plants per plot). Greenhouse temperature and relative humidity were maintained at approximately 28°C and 60%–80%. Plants were fertigated with Yamazaki nutrient solution (EC 2.8 dS/m, pH 6.0) through drip irrigation supplied at 20-min intervals between 07:30 and 16:30.
To ensure statistical representativeness and minimize intra-plot variability, three uniform plants were selected from each replicate, resulting in nine plants per treatment (n = 9) being used as the experimental units for all vegetative and reproductive parameters. Vegetative growth was assessed through plant height, stem diameter, growth-zone diameter (10 cm below the flowering truss), leaf number, and leaf blade dimensions was measured every 2 weeks. Leaf area was estimated using the regression model of Carmassi et al.(2007):
Leaf area (cm2) =-10.12 + 0.834 × (0.5 × leaf length×leaf width)
Total leaf area per plant was calculated as the cumulative sum of measured leaves. Reproductive development was assessed by counting flower buds, open flowers, and fruit set for each inflorescence. Early yield was measured from the first two trusses; fruit number, fresh weight, and mean fruit weight were recorded. Yield per plant (g/plant) was converted to area-based yield (t/ha) using planting density.
2.3. Statistical analysis
All statistical analyses were performed using SAS 9.4 (SAS Institute Inc., Cary, NC, USA). To specifically evaluate the concentration-dependent effects of diniconazole at each developmental stage, data collected at different time intervals (WAS and WAT) were subjected to a one-way analysis of variance (ANOVA) independently. Mean separation was conducted using Tukey’s multiple range test at P < 0.05. Figures were prepared with SigmaPlot 12.5 (Systat Software Inc., San Jose, CA, USA).
3. Results and Discussion
3.1. Effects of diniconazole application on seedling quality during the nursery stage
Diniconazole concentration based leaf development was observed at each stage of seedling growth (Table 1). At 4 weeks after sowing (WAS), the highest concentration (250 mg/L) markedly reduced leaf area, consistent with the role of triazole growth regulators in suppressing gibberellin-mediated cell expansion (Rademacher, 2000). In contrast, 50 and 125 mg/L produced leaf areas comparable to the control. As seedlings progressed into mid-development (5 WAS), treatment wise variability was not pronounced, implying a temporary compensatory growth phase.
Effects of diniconazole concentration on leaf morphological traits and chlorophyll content (SPAD value) of tomato seedlings at different growth stages, raised in a growth chamber at 25°C
During the late nursery stage (6–7 WAS), untreated seedlings exhibited strong reductions in leaf area and premature leaf senescence, likely due to rapid substrate nutrient depletion associated with excessive early vegetative growth. In contrast, diniconazole-treated seedlings maintained greater leaf area, with the 125 mg/L treatment producing the highest leaf area and leaf number. Chlorophyll content followed a similar pattern: SPAD values of untreated seedlings declined steadily, while all diniconazole treatments maintained SPAD values above 50. The enhanced chlorophyll retention, particularly at 125 mg/L, aligns with previous findings that triazoles delay chlorophyll degradation through suppression of GA signaling and protection of chloroplast integrity (Fletcher et al., 2000).
Overall, diniconazole suppressed early leaf expansion but enhanced physiological stability later in the nursery stage.
Diniconazole significantly suppressed shoot elongation in a concentration-dependent manner (Table 2; Fig. 1). Height reduction was the primary sign due to shortened internodes, which is consistent with the inhibition of GA biosynthesis, particularly the suppression of ent-kaurene oxidation by triazole compounds (Davis et al., 1988; Rademacher, 2000). As the height suppressed, stem thickening progressed from 5 WAS onwards, especially at 125 mg/L, resulting in compact seedlings.

Effects of diniconazole concentrations on number of nodes, stem diameter, plant height, and root length of tomato seedlings at different growth stages, raised in a growth chamber at 25°C

Representative morphological responses of tomato seedlings raised in a plant growth chamber maintained at 25°C under different diniconazole concentrations (0, 50, 125, and 250 mg/L) from 3 to 7 weeks after treatment.
The shift in assimilate allocation from longitudinal to radial growth reinforces that triazole-induced height control redirects photoassimilates to supportive tissues (Fletcher et al., 2000; Desta and Amare, 2021). By 7 WAS, seedlings treated with 125 mg/L developed more nodes despite being shorter, confirming that the triazole compound reduced internode length without impairing meristematic activity (Jang et al., 2020).
Root growth at a concentration of 125 mg/L was comparable to the control, indicating that moderate diniconazole application did not compromise root system development. This shoot–root balance is agronomically desirable because it enhances post-transplant establishment (Latimer, 1992). Excessive suppression at 250 mg/L, however, diminished overall vigor, likely due to restricted assimilate production and impaired structural development (Choi et al., 2001).
Thus, diniconazole at 125 mg/L effectively suppressed internode elongation while preserving stem diameter and root development, yielding compact, vigorous seedlings ideal for transplanting.
Biomass accumulation was strongly effected by diniconazole concentration (Table 3). At 4 WAS, high-concentration application (250 mg/L) significantly reduced biomass, indicating that excessive suppression of vegetative growth limited assimilate production. As seedlings matured, the 125 mg/L treatment exhibited superior biomass accumulation. By 6 WAS, total fresh weight was approximately one-third greater than the control, and root fresh weight consistently exceeded that of untreated seedlings. By 7 WAS, dry matter accumulation was highest at 125 mg/L, suggesting enhanced tissue density and improved dry matter partitioning.

Effects of diniconazole concentrations on fresh weight, and dry weight of tomato seedlings at different growth stages, raised in a growth chamber at 25°C
These results are consistent with existing studies showing that moderate triazole application increases tissue density and mechanical strength through shifts in assimilate allocation (Fletcher et al., 2000; Niu et al., 2002). In contrast, substantial growth suppression at 250 mg/L, resulted in the lowest biomass throughout development, emphasizing the risk of excessive dosage.
Overall, diniconazole at 125 mg/L resulted in structural robustness and biomass balance in tomato seedlings. Consequently, these seedlings exhibited compact shoot and root growth with sustained vigor, ensuring high transplant quality.
3.2. Carry-over effects of diniconazole on early growth and yield after transplanting
Carry-over effects of diniconazole applied during the nursery stage were evident immediately after transplanting and persisted through early vegetative stage (Table 4). At 1 week after transplanting (WAT), control plants exhibited the smallest leaf area and lowest SPAD values, likely due to the rapid shift toward stem elongation that restricted leaf expansion and photosynthetic capacity. In contrast, seedlings previously treated with 125 mg/L produced significantly greater leaf area and maintained stable SPAD values above 50, indicating superior photosynthetic potential and more stable early growth. These results support findings that moderate height suppression during seedling production improves post-transplant vigor by conserving assimilates for later leaf development (Vavrina and Orzolek, 1993).

Effects of seedling-stage diniconazole treatments on leaf growth and chlorophyll content of tomato plants at different growth stages after transplanting in a greenhouse
From 3 to 5 WAT, all treatments showed increases in leaf number and area; however, diniconazole-treated plants consistently exhibited higher SPAD values, reflecting delayed chlorophyll degradation and enhanced leaf physiological stability. The 125 mg/L treatment produced more leaves and maintained higher chlorophyll content than the control while preventing excessive canopy enlargement. This pattern aligns with the known triazole-mediated reductions in internode elongation and leaf enlargement that promote efficient assimilate allocation to functional leaf tissues (Yun et al., 2007).
By 13 WAT, differences among the control, 50 mg/L, and 125 mg/L treatments had largely diminished, suggesting that these concentrations did not impose long-term restrictions on leaf growth or chlorophyll content. In contrast, the 250 mg/L treatment resulted in decreased leaf number, leaf area, and SPAD values, suggesting impaired leaf development and photosynthetic competence caused by prolonged inhibition of GA biosynthesis (Rademacher, 2000).
Morphological differences established during the nursery period persisted after transplanting and continued to influence stem elongation, radial thickening, and node formation (Table 5; Fig. 2). At 1 WAT, control plants showed pronounced stem elongation, whereas all diniconazole-treated plants remained significantly shorter (23.0–25.5 cm). This persistent height suppression reflects the earlier inhibition of internode extension caused by triazole-mediated GA biosynthesis reduction (Rademacher, 2000).

Effects of seedling-stage diniconazole treatments on number of nodes, basal stem diameter, apical stem diameter, and plant height of tomato plants at different growth stages after transplanting in a greenhouse

Morphological characteristics of tomato plants at 13 weeks after transplanting as influenced by different diniconazole concentrations applied during the seedling stage.
By 3 WAT, plants treated with 50 and 125 mg/L had heights similar to the control, but stem diameter remained consistently greater in all diniconazole treatments, indicating sustained allocation of assimilates toward radial growth rather than elongation. Such thickened stems are advantageous for structural strength and hydraulic conductivity, supporting improved long-term vigor.
During the mid-season (5–7 WAT), differences became more pronounced. Plants treated with 250 mg/L developed the thickest stems but remained significantly shorter than all other treatments, reflecting cumulative GA inhibition and reduced metabolic activity, a response previously reported under excessive triazole exposure (Jang et al., 2020). At 13 WAT, the control plants exhibited the greatest height, while the 125 mg/L treatment maintained a compact and agronomically desirable canopy. Node number did not differ among the control, 50 mg/L, and 125 mg/L treatments, indicating that moderate concentrations affected internode elongation but not meristematic activity. Conversely, the 250 mg/L treatment produced fewer nodes, suggesting suppressed meristem function under excessive dosage.
Analogous to nursery stage, 125 mg/L treatment provided the most favorable response, supporting compact growth with enhanced stem diameter and stable node development traits that contribute to improved performance in greenhouse cultivation.
Carry-over effects of diniconazole on reproductive development were substantial and highly concentration dependent (Table 6). At 3 WAT, all diniconazole treatments showed accelerated floral transition compared to the control, with increased numbers of floral primordia and open blooms. The 250 mg/L diniconazole treatment yielded the highest count of early floral structures, indicating that intense vegetative suppression in seedlings diminished competing sinks and promoted earlier reproductive onset, a pattern aligning with growth regulator effects in compact transplants (Zhang et al., 2003).

Effects of seedling-stage diniconazole treatments on number of flowers, number of blooms, and number of fruit sets of tomato plants at different growth stages after transplanting in a greenhouse
Despite this early stimulation, fruit-set efficiency varied widely. Although the 250 mg/L treatment produced many early flower buds, it had relatively low fruit-set rates, while the 50 mg/L treatment achieved the highest fruit set rate at 3 WAT. This indicates that excessive triazole application may reduce assimilate production and transport, limiting the conversion of flowers to fruit, a constraint consistent with known metabolic effects of high triazole concentrations (Rademacher, 2000).
During mid-season (5–7 WAT), fruit set increased across all diniconazole-treated plants producing 2–2.5 times more fruit than the control. The 125 mg/L treatment consistently produced the highest fruit-set numbers at 7 WAT, ascertaining a sustained reproductive advantage beyond the early establishment stage. In contrast, the 250 mg/L treatment did not maintain its initial reproductive stimulation; despite producing abundant floral primordia early, fruit-set retention declined, likely due to limited assimilate supply and cumulative metabolic suppression.
By 9 WAT, treatment effects were fully expressed. Plants treated with 125 mg/L maintained the greatest fruit-set count (14.8 fruits per plant), indicating strong reproductive continuity and sufficient assimilate supply (Yun et al., 2007). Conversely, the 250 mg/L treatment exhibited a major decline in fruit set, reflecting long-term constraints on carbon assimilation and resource allocation (Choi et al., 2001).
Overall, diniconazole imposed distinct responses to various concentrations, during reproductive phase post-transplanting. Moderate application rates enhanced early floral initiation, supported stable mid-season reproductive activity, and ensured sustained fruit-set retention during prolonged greenhouse cultivation. Excessive application (250 mg/L), while stimulating early floral differentiation, ultimately reduced fruit-set efficiency and long-term reproductive performance.
Early yield and harvest earliness were markedly influenced by diniconazole application during the seedling stage, and responses varied strongly with treatment concentration (Table 7; Fig. 3). Among all treatments, 125 mg/L produced the most favorable carry-over effects, leading to the earliest harvest initiation and the greatest cumulative early yield. Yield from the first two trusses reached 663.4 g/plant (20.7 t/ha), representing a 75% increase relative to the untreated control (379.3 g/plant; 11.9 t/ha) which was statistically significant (P < 0.05) (Table 7; Fig. 4). These findings indicate that moderate suppression of vegetative vigor during the nursery stage enhances assimilate partitioning toward reproductive sinks after transplanting, thereby improving early-fruiting performance. Similar improvements in early yield resulting from compact seedling architecture have been reported in tomato and other solanaceous crops (Marcelis, 1996; Kim et al., 2016).

Effects of seedling-stage diniconazole treatments on cumulative fruit number per plant, cumulative fruit weight, mean fruit weight, and estimated yield per hectare of tomato plants at 13 weeks after transplanting under greenhouse conditions
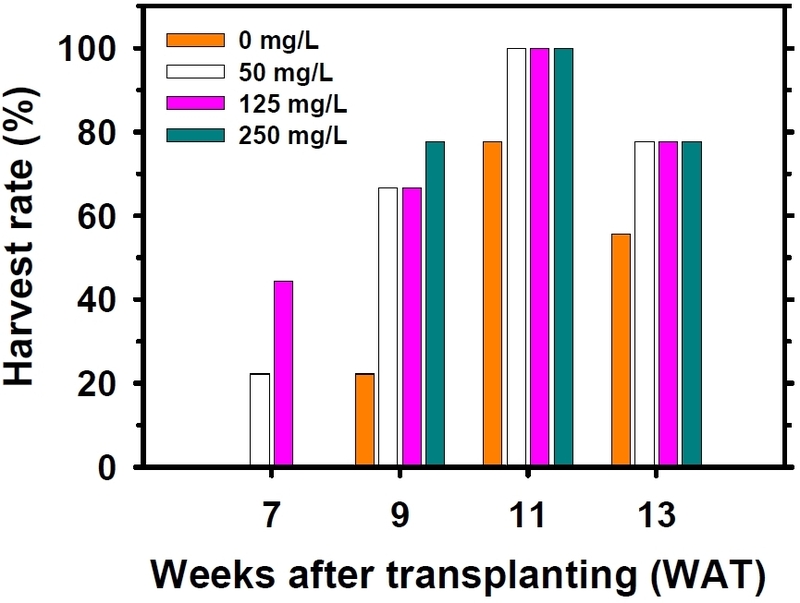
Changes in harvest rate of greenhouse-grown tomato plants as influenced by diniconazole concentrations applied during the seedling stage. Harvest rate was determined at 2-week intervals and expressed as the percentage of plants producing marketable fruits relative to the total number of plants per treatment (n = 9).
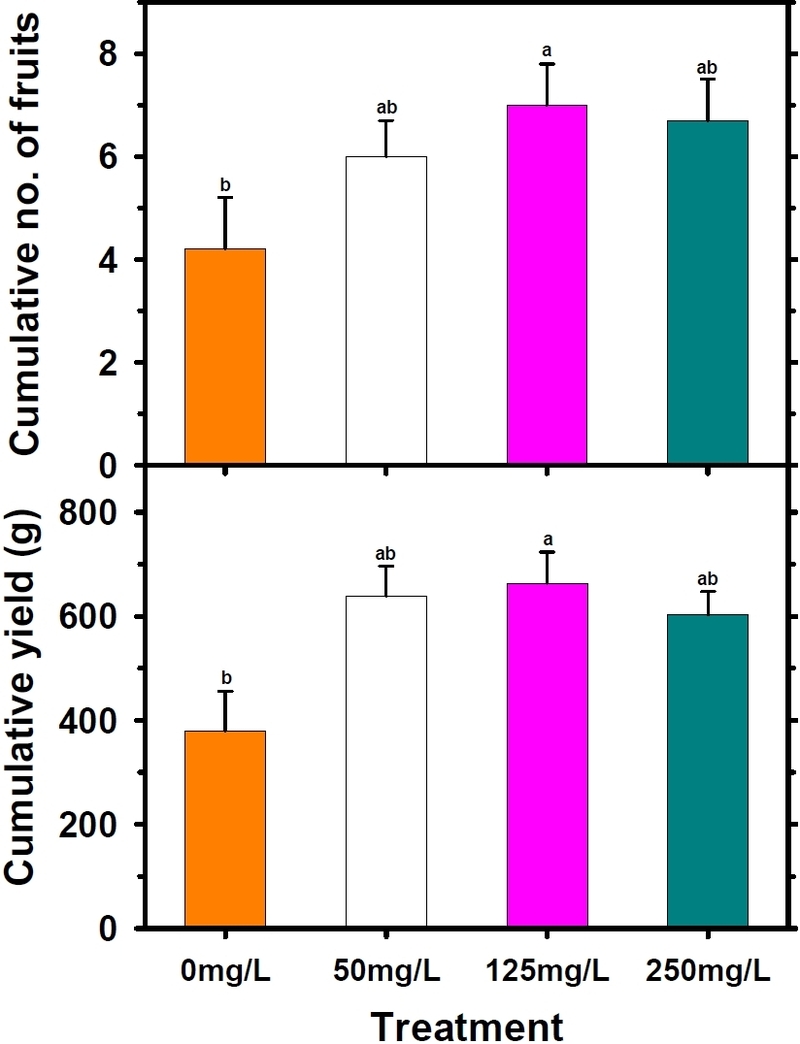
Effects of diniconazole treatments applied during the seedling stage on cumulative fruit number and cumulative yield of tomato plants grown in a greenhouse. Cumulative fruit number and yield were determined from the first and second trusses. Error bars represent the standard error of the mean (n=9).
Although the 250 mg/L treatment produced the highest number of initial fruit sets, its early yield (18.8 t/ha) was lower than that of the 125 mg/L treatment because of reduced mean fruit weight. This responses suggests analogy to previous reports that excessive triazole-induced growth inhibition may accelerate floral induction while restricting assimilate availability during fruit enlargement, thereby reducing final fruit size despite abundant fruit set (Marcelis, 1996; Jang et al., 2020). Across treatments, mean fruit weight varied only modestly (84.9–102.9 g); thus, differences in early yield were primarily driven by variation in fruit set and the timing of harvest onset.
Weekly harvest patterns (Fig. 3) further confirmed the benefits of moderate diniconazole application. Plants treated with 50 and 125 mg/L initiated harvest 2 weeks earlier than both the control and the 250 mg/L treatment. Notably, the 125 mg/L treatment produced the highest proportion (44%) of marketable fruits during week 7 after transplanting, indicating enhanced precocity. These results are consistent with the hypothesis that compact seedlings exhibit greater early reproductive sink strength and more efficient allocation of assimilates to fruit development, particularly during the initial post-transplant period (Talukder et al., 2025).
In contrast, the reduced early yield of the 250 mg/L treatment suggests that excessive inhibition of vegetative growth compromises the sustained supply of assimilates required for early fruit enlargement, even when flower initiation is promoted. Such negative impacts of high triazole concentrations on reproductive productivity have been documented in several horticultural crops (Rademacher, 2000; Jang et al., 2020).
Overall, these results demonstrate that diniconazole application during the seedling stage exerts substantial concentration-dependent carry-over effects on early tomato productivity. Among the treatments examined, 125 mg/L represents to be ideal concentration, simultaneously enhancing harvest earliness and maximizing early cumulative yield.
4. Conclusions
This study reveals that diniconazole application at the tomato seedling stage markedly enhances transplant quality and early-season performance. Treatments suppressed plant height and internode elongation in a concentration-dependent manner, with 125 mg/L yielding optimal seedlings characterized by sustained chlorophyll content, balanced shoot–root growth, and improved morphological compactness. Post-transplant, these plants showed earlier fruit set, harvest initiation, and significantly higher early yield per unit area compared to controls, driven primarily by increased fruit number rather than size. Conversely, 250 mg/L induced excessive inhibition, elevating floral counts but restricting fruit enlargement and total yield. Despite these significant findings, certain limitations still exist in this study such as the results depicted only the early yield factors, while yield assessment for whole crop cycle is yet to be studied. Further physiological investigations involving direct measurements of gas exchange and carbon partitioning are would elucidate the precise mechanisms underlying the observed carry-over effects.
Nevertheless, These results demonstrate that 125 mg/L diniconazole effectively modulates seedling vigor to boost tomato productivity, indicating that appropriate modulation of seedling growth using triazole-based regulators can serve as an effective strategy for optimizing transplant quality and early-season productivity in tomato production systems.
Acknowledgments
This work was carried out with the support of “Cooperative Research Program for Agriculture Science and Technology Development (Project No. RS-2025-02303116)”, Rural Development Administration, Korea.
REFERENCES
-
Ali, M. R., Quddus, M. A., Trina, T. N., Salim, M. M. R., Asaduzzaman, M., 2022, Influence of plant growth regulators on growth, yield, and quality of tomato grown under high temperature in the tropics in the summer, Int. J. Veg. Sci., 28, 59-75.
[https://doi.org/10.1080/19315260.2020.1828526]

-
Blanchard, M. G., Runkle, E. S., 2007, Dipping bedding plant liners in paclobutrazol or uniconazole inhibits subsequent stem extension, HortTech., 17, 178-182.
[https://doi.org/10.21273/HORTTECH.17.2.178]

- Choi, Y. H., Rhee, H. C., Park, D. K., Kwon, J. K., Lee, J. H., 2001, Effects of mechanical stimulation and chemical treatments on growth of seedlings and yield of tomato, Hortic. Sci. Technol., 19, 320-324.
-
Davis, T. D., Steffens, G. L., Sankhla, N., 1988, Triazole plant growth regulators, in: Janick, J. (ed.), Horticultural Reviews, vol. 10, 1st ed., Wiley-Blackwell, Hoboken, USA, 63-105.
[https://doi.org/10.1002/9781118060834.ch3]

-
Desta, B., Amare, G., 2021, Paclobutrazol as a plant growth regulator, Chem. Biol. Technol. Agric., 8, 1.
[https://doi.org/10.1186/s40538-020-00199-z]

-
Dunn, B. L., Goad, C., Brandenberger, L., 2022, Growth and flowering of greenhouse-grown tomato transplants in response to uniconazole, HortTech., 32, 485-490.
[https://doi.org/10.21273/HORTTECH05071-22]

-
Fan, X. X., Xu, Z. G., Liu, X. Y., Tang, C. M., Wang, L. W., Han, X. L., 2013, Effects of light intensity on the growth and leaf development of young tomato plants grown under a combination of red and blue light, Sci. Hortic., 153, 50-55.
[https://doi.org/10.1016/j.scienta.2013.01.017]

-
Fletcher, R. A., Gilley, A., Sankhla, N., Davis, T. D., 2000, Triazoles as plant growth regulators and stress protectants, in: Janick, J. (ed.), Horticultural Reviews, vol. 24, 1st ed., Wiley-Blackwell, Hoboken, USA, 55-138.
[https://doi.org/10.1002/9780470650776.ch3]

-
Heins, R. D., Liu, B., Runkle, E. S., 1998, Regulation of crop growth and development based on environmental factors, Acta Hortic., 513, 17-28.
[https://doi.org/10.17660/ActaHortic.1998.513.1]

-
Jang, D. C., Xu, C., Kim, S. H., Kim, D. H., Kim, J. K., Heo, J. Y., Vu, N. T., Choi, K. Y., Kim, I. S., 2020, Effects of different application approaches with diniconazole on the inhibition of stem elongation and the stimulation of root development of cylindrical paper pot seedling, Prot. Hortic. Plant Fact., 29, 365-372.
[https://doi.org/10.12791/KSBEC.2020.29.4.365]

-
Kim, H. C., Cho, Y. H., Ku, Y. G., Hwang, S. J., Bae, J. H., 2016, Growth characteristics of grafted tomato seedlings following treatment with various concentrations of diniconazole during the summer growth season, Hortic. Sci. Technol., 34, 249-256.
[https://doi.org/10.12972/kjhst.20160026]

-
Kubota, C., McClure, M. A., Kokalis-Burelle, N., Bausher, M. G., Rosskopf, E. N., 2008, Vegetable grafting: History, use, and current technology status in North America, HortSci., 43, 1664-1669.
[https://doi.org/10.21273/HORTSCI.43.6.1664]

-
Latimer, J. G., 1992, Drought, paclobutrazol, abscisic acid, and gibberellic acid as alternatives to daminozide in tomato transplant production, J. Am. Soc. Hortic. Sci., 117, 243-247.
[https://doi.org/10.21273/JASHS.117.2.243]

-
Marcelis, L. F. M., 1996, Sink strength as a determinant of dry matter partitioning in the whole plant, J. Exp. Bot., 47, 1281-1291.
[https://doi.org/10.1093/jxb/47.Special_Issue.1281]

-
Myster, J., Moe, R., 1995, Effect of diurnal temperature alternations on plant morphology in some greenhouse crops—a mini review, Sci. Hortic., 62, 205-215.
[https://doi.org/10.1016/0304-4238(95)00783-P]

-
Rademacher, W., 2000, Growth retardants: Effects on gibberellin biosynthesis and other metabolic pathways, Annu. Rev. Plant Physiol. Plant Mol. Biol., 51, 501-531.
[https://doi.org/10.1146/annurev.arplant.51.1.501]

-
Schwarz, D., Rouphael, Y., Colla, G., Venema, J. H., 2010, Grafting as a tool to improve tolerance of vegetables to abiotic stresses: Thermal stress, water stress and organic pollutants, Sci. Hortic., 127, 162-171.
[https://doi.org/10.1016/j.scienta.2010.09.016]

-
Talukder, M. R., et al., 2025, Fluctuation of ambient day-night temperature influences morphological traits, floral characters, fruit yield and quality of summer tomato genotypes grown in hydroponics, N. Z. J. Crop Hortic. Sci., 1-24.
[https://doi.org/10.1080/01140671.2025.2504209]

-
Vavrina, C. S., Orzolek, M. D., 1993, Tomato transplant age: A Review, HortTech., 3, 313-316.
[https://doi.org/10.21273/HORTTECH.3.3.313]

- Yun, H. K., Seo, T. C., Lee, J. W., Yang, E. Y., 2007, Effect of triazole growth regulator treatment on the growth of plug seedling and yield of tomato, J. Bio-Environ. Control, 16, 205-209.
- Zhang, C. H., Chun, I. J., Park, Y. C., Kim, I. S., 2003, Effect on the inhibition of over-growth of plug seedling by triazole-type growth regulator treatment, J. Bio-Environ. Control, 12, 139-146.
Department of Horticultural Bioscience, Pusan National Universitykangjs@pusan.ac.kr
Department of Horticultural Bioscience, Pusan National Universityjaidergo@naver.com
Department of Horticultural Bioscience, Pusan National Universitytkdflajaid@naver.com
Department of Horticultural Bioscience, Pusan National Universitymdfaraazbio1803@gmail.com
Department of Horticultural Bioscience, Pusan National Universitydlwlrn15@naver.com
Department of Horticultural Bioscience, Pusan National Universitymcchryl@gmail.com
Department of Horticultural Bioscience, Pusan National Universitymg6188@naver.com
Department of Horticultural Bioscience, Pusan National Universitydaegeunjeong99@gmail.com
Department of Horticultural Bioscience, Pusan National Universityatom0821@pusan.ac.kr