Physicochemical Characterization of Soluble and Insoluble Dietary Fiber Fractions from Apple Pomace Using Green Pre-treatment: Toward Carbon Reduction and Circular Resource Valorization
Ⓒ The Korean Environmental Sciences Society. All rights reserved.
This is an Open-Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
In South Korea, apple processing generates substantial amounts of apple pomace (AP), a moisture-rich byproduct that can threaten the environment if not managed promptly. Because AP is rich in dietary fiber but predominantly insoluble, increasing the soluble dietary fiber (SDF) fraction can expand its potential for higher-value upcycling applications. Although SDF recovery often relies on strong-acid extraction, eco-friendly pre-treatment strategies may reduce chemical inputs and downstream treatment demands, thereby supporting circular resource valorization. To fractionate AP into SDF and insoluble dietary fiber (IDF), an untreated control, an HCl-assisted extraction route, and fermentation pre-treatment with Aspergillus luchuensis 74-5 were compared. The SDF and IDF fractions were recovered after extraction and ethanol precipitation, and their yield, hydration, and dispersion properties (solubility, water absorption, and dispersibility) were measured. Structural features were assessed using spectroscopic, chromatographic, diffraction, and microscopic analyses. Fermentation increased the SDF yield from 1.51% to 10.17%, approaching that of HCl-assisted extraction (12.00%), while maintaining a higher IDF yield than that of HCl-assisted extraction (p < 0.05). The 74-5–derived SDF fraction showed higher solubility (93.19%) and lower water absorption (0.80 g/g) than the HCl-derived fraction (49.39% and 2.60 g/g), with comparable dispersibility. Structural analyses revealed workflow-dependent differences in polymer distribution and microstructure, which were consistent with the observed hydration and dispersion properties. Overall, these results suggest that the 74-5 fermentation–based workflow can support circular resource valorization of AP while potentially reducing chemical inputs and downstream treatment burdens compared with acid-assisted extraction.
Keywords:
Apple pomace, Circular resource valorization, Aspergillus luchuensis, Fermentation, Hot-water extraction, Ethanol precipitation, Soluble dietary fiber1. Introduction
Apple processing generates large volumes of apple pomace (AP), a by-product remaining after juice and cider production (Costa et al., 2022; Thomas et al., 2025). Improperly managed landfilling of AP and other biodegradable fruit-processing residues can generate methane emissions and leachate (Czubaszek et al., 2022; Putra et al., 2023). In South Korea, apple production remains substantial, with recent estimates placing national output at roughly 0.43 million metric tons and orchard area at around 33,900 ha (USDA Foreign Agricultural Service (FAS), 2023; Korea Agro–Fisheries & Food Trade Corporation (aT), 2023). From an environmental-science perspective, upcycling—converting low-value byproducts into higher-value products—can reduce disposal impacts and potentially lower waste–management–related greenhouse-gas emissions (Ampese et al., 2023; Azarakhsh et al., 2024), while enabling the recovery of bioactive compounds and biopolymer-rich fractions from agri-food by-products (Ben-Othman et al., 2020; Pascoalino et al., 2025).
AP is generated in large quantities but is often underutilized because its high moisture content and compositional heterogeneity accelerate spoilage and complicate collection, storage, and transport; therefore, rapid stabilization and careful management are required (Vandorou et al., 2024; Thomas et al., 2025). Nevertheless, systematic reviews and meta-analyses support incorporating AP for food fortification across diverse food categories, underscoring its potential as a value-added valorization pathway aligned with waste-management objectives (Antonic et al., 2020; Lyu et al., 2020; Zaky et al., 2024). In this context, fractionating AP into standardized, functionality-oriented ingredient streams—such as soluble and insoluble dietary-fiber fractions (SDF and IDF)—can enhance value capture and broaden application potential by providing more consistent, fit-for-purpose materials (Buljeta et al., 2023; Thomas et al., 2025).
AP is increasingly recognized as a fiber-rich by-product for value-added utilization because it contains abundant cell-wall polysaccharides such as cellulose, hemicellulose, and pectin; however, its high moisture content and perishability necessitate prompt stabilization (e.g., drying) to enable storage and downstream processing (Vandorou et al., 2024; Thomas et al., 2025). To obtain standardized dietary-fiber fractions—particularly soluble dietary fiber (SDF) for incorporation into food matrices—enhancing partial solubilization of the cell-wall matrix is a key step (Buljeta et al., 2023; Al Faruq et al., 2025). Acid-assisted extraction (e.g., HCl) can promote solubilization, but conventional acid– and base-assisted routes may generate chemical wastes and acidic or alkaline effluents requiring neutralization and treatment prior to discharge and may contribute to equipment corrosion, increasing operational and environmental burdens (Riyamol et al., 2023; Pattarapisitporn and Noma, 2025). Therefore, low-chemical pre-treatment strategies may offer carbon- and resource-efficiency benefits (Riyamol et al., 2023; Pattarapisitporn and Noma, 2025); workflow selection and optimization for targeted SDF and IDF recovery are largely governed by extraction methods and key processing conditions (temperature, pH, solvent, time, and liquid–to–solid ratio) (Buljeta et al., 2023; Kaur et al., 2025).
A range of pre-treatment and modification strategies (chemical and enzymatic treatments, thermomechanical processing, and microbial fermentation) has been explored to enrich SDF and improve fiber functionality, as these routes can reshape fiber microstructure and physicochemical properties, with increasing interest in more sustainable processing (Liu et al., 2024; Kumari et al., 2025; Zhao et al., 2025). In particular, sustainability-oriented “green” approaches can improve SDF yield and functionality (Al Faruq et al., 2025; Zhao et al., 2025). Within this framework, fermentation-based pre-treatment is a promising option because it leverages microbial enzymes to partially disassemble the cell-wall matrix, thereby promoting SDF release (Wang et al., 2024a; Al Faruq et al., 2025). However, several emerging green technologies—including microwave, ultrasound, and high-pressure homogenisation—remain costly and challenging to scale up (Al Faruq et al., 2025).
As a food-grade koji mold rich in carbohydrate-active enzymes, Aspergillus luchuensis 74-5 can partially deconstruct plant cell-wall polysaccharides to increase extractable soluble fractions, potentially supporting SDF enrichment during downstream fractionation (Yamashita, 2021; Futagami, 2022; Li et al., 2024). However, AP valorization studies often focus on isolated unit operations, with limited benchmarking of end-to-end workflows or clear links between process selection and the physicochemical performance of the resulting SDF and IDF ingredients (Asif et al., 2024; Thomas et al., 2025).
This gap highlights the need for a practical pre-treatment-to-fractionation strategy that (i) increases SDF recovery to improve processability and incorporation in aqueous formulations (Al Faruq et al., 2025; Kumari et al., 2025), (ii) preserves IDF yield to retain overall fiber mass and co-product potential (Ma et al., 2025a; Mammolenti et al., 2025), and (iii) reduces chemical inputs and downstream treatment requirements in support of circular-resource valorization (Al Faruq et al., 2025; Pascoalino et al., 2025).
Accordingly, this study compares a green workflow (A. luchuensis 74-5 fermentation → hot-water extraction → ethanol precipitation) with an untreated control and an HCl-assisted extraction route. Outcomes assessed include SDF and IDF yields, hydration and dispersion properties (solubility, water absorption, dispersibility), and structural signatures (FT-IR, GPC, XRD, SEM, TEM). Collectively, these comparisons clarify how process selection drives fraction recovery and functional performance, informing sustainable, circular valorization of apple pomace.
2. Materials and Methods
2.1. Apple pomace
Apple pomace (AP) was obtained from Chungcheongbuk-do Horticulture Nonghyup (Chungcheongbuk-do, Republic of Korea). The pomace was derived from Fuji apples and comprised peel, seeds, and residual pulp remaining after mechanical juice pressing. The AP was transported to the laboratory frozen and stored at −20℃ until further use.
2.2. Reagents
Ethanol was purchased from Ethanol Supplies World Co., Ltd. (Jeonju-si, Republic of Korea). A commercial white-koji starter culture containing Aspergillus luchuensis 74-5 (Suwon Jongguk) was obtained from Suwon Fermentation Co., Ltd. (Hwaseong-si, Republic of Korea) and used for fermentation pre-treatment.
2.3. Experimental design and pre-treatment conditions
AP stored at −20℃ was thawed at 4℃ prior to treatment, then divided into three groups (Control, 74-5, and HCl), with all experiments performed in triplicate (n = 3). For the Control and HCl groups, thawed AP was incubated at room temperature (RT; 20-25℃) for 7 days without starter addition. For fungal pre–treatment (74-5 group), AP was mixed with a commercial white–koji starter culture containing Aspergillus luchuensis 74-5 at 1% (w/w) and incubated at RT for 7 days under the same time and temperature conditions. After incubation, all samples were hot-air dried at 50℃ to constant weight prior to extraction and fractionation (Section 2.4), with the HCl group subsequently subjected to acid-assisted extraction as described below.
2.4. Extraction and fractionation
A schematic overview of the extraction and fractionation into SDF and IDF is shown in Fig. 1. SDF and IDF fractions were separated from the pre-treated, dried AP following Szymańska-Chargot et al.(2017) with minor modifications. Briefly, dried AP (150 g) was extracted at 85℃ for 30 min per cycle (two cycles total) using distilled water for the Control and 74-5 groups (1.5 L per cycle) or hydrochloric acid solutions for the HCl group (1 M for 30 min followed by 0.5 M for 30 min). After each extraction cycle, the slurry was vacuum-filtered through Whatman Grade 4 filter paper (20-25 μm particle retention), and the retained residue was collected as the IDF fraction. Filtrates from both cycles were pooled and mixed with ethanol at a filtrate:ethanol ratio of 1:4 (v/v), followed by precipitation at 4℃ for 24 h. The precipitate was recovered by vacuum filtration as the SDF fraction. Both SDF and IDF fractions were hot-air dried at 50℃ to constant weight, ground into powders, and used for subsequent analyses.
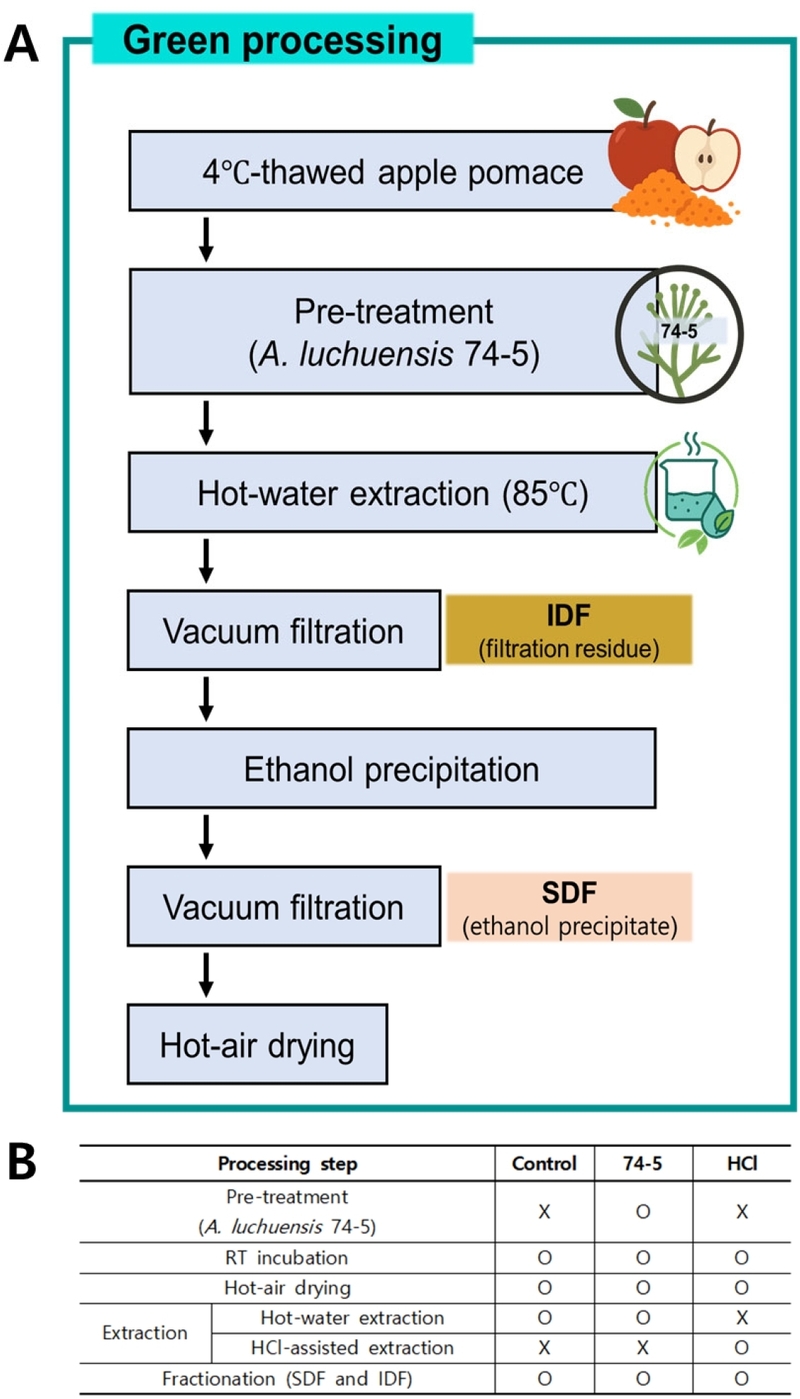
(A) Schematic workflow for SDF and IDF fractionation from apple pomace highlighting the green (74-5) process, and (B) summary of steps applied in the Control, 74-5, and HCl treatments. (A) provides a schematic of the green (A. luchuensis 74-5) process for SDF and IDF fractionation: IDF was collected as the filter-retained residue after vacuum filtration, and SDF was recovered from pooled filtrates by ethanol precipitation followed by vacuum filtration. (B) summarizes the processing steps applied to each treatment group (Control, 74-5, and HCl); ○, applied; ×, not applied; RT, room temperature (20–25℃).
2.5. Physicochemical properties
Yields were calculated on a dry-weight basis after hot-air drying at 50℃ to constant weight. Overall yield (%) was calculated as:
SDF or IDF yield (%) = (dry extract mass, g × 100) / (pre-treated dried sample mass, 150 g).
Water solubility was evaluated using the water solubility index (WSI) with minor modifications of the method described by Sifat et al.(2021). 1.0 g of powder (<212 μm) was mixed with 12 mL of distilled water in a 50 mL centrifuge tube and stirred at 30℃ for 30 min. The mixture was centrifuged (3,000 rpm, 30 min, 4℃). The supernatant was carefully collected and oven-dried at 105℃ until constant weight (≈4 h) to obtain the mass of dissolved solids. WSI (%) was calculated as:
WSI (%) = (mass of solids in the dried supernatant, g × 100) / (dry sample mass, 1.0 g).
Water absorption was determined using the water absorption index (WAI) based on the wet pellet remaining after the WSI test. After removing the supernatant, the wet pellet was weighed, and WAI (g/g) was calculated as:
WAI (g/g) = (wet pellet mass after centrifugation, g) / (dry sample mass, 1.0 g).
Dispersibility was evaluated with minor modifications of the method described by Jinapong et al.(2008). A 1.0 g of powdered sample was mixed with 10 mL of distilled water in a 50 mL centrifuge tube and vortexed for 20 s to form a suspension. The suspension was immediately filtered through a 212 μm stainless-steel sieve. An aliquot of the filtrate (1.0 mL) was transferred to a pre-weighed aluminum dish and dried in an oven at 105℃ for 4 h to constant weight to determine total solids (%TS). %TS was calculated as:
%TS = (mass of dried solids after drying at 105℃, g × 100) / (mass of filtrate, 1.0 g)
Dispersibility (%) was calculated as:
Dispersibility (%) = ((10 + a) × %TS) / (a × ((100 − b) /100))
where a is the mass of the dry sample added (g; 1.0 g in this study), b is the initial moisture content of the dry sample (%; 0% in this study), and 10 represents the mass of water added (10 mL ≈ 10 g).
2.6. Structural and morphological characterization
Structural and morphological analyses (FT-IR, GPC, XRD, SEM, and TEM) were performed at the Korea Polymer Testing & Research Institute (KOPTRI, Seoul, Republic of Korea). Measurement conditions are described below.
FT-IR spectra were acquired using a JASCO FT/IR-4600 spectrometer equipped with an ATR accessory (Ge crystal). Spectra were recorded over 4,000-700 cm⁻¹ with 32 scans at a resolution of 4 cm⁻¹.
Molecular-weight distribution was determined by gel permeation chromatography (GPC) using a Tosoh HLC-8420 system equipped with a refractive index (RI) detector. The separation was performed under the size-exclusion principle, where analytes are resolved primarily by hydrodynamic size (larger species elute earlier). Samples (3 mg/mL) were dissolved in 0.1 M NaNO₃, filtered through a 0.22 μm nylon membrane filter, and injected with a 50 μL injection volume. Separation was performed using a column set consisting of a TSKgel guard PWxl, two GMPWxl columns, and a G2500PWxl column maintained at 40℃. The mobile-phase flow rate was 1.0 mL/min. Molecular weights were calibrated using PEG/PEO standards, and chromatographic data were processed with EcoSEC Elite-WS software.
XRD patterns were collected using a Bruker D8 ADVANCE diffractometer with Cu Kα radiation (40 kV, 40 mA). Data were recorded over a 2θ range of 5–90°.
Surface morphology was examined using a Hitachi SU5000 field-emission scanning electron microscope (FE-SEM). Samples were platinum–coated (F-Pt-6-200 condition) prior to imaging. Micrographs were acquired at an accelerating voltage of 5 kV, beam intensity of 30, and magnifications of ×200, ×1,000, and ×10,000. The instrument resolution was 1.2 nm at 30 kV and 3.0 nm at 1 kV.
Ultrastructural features were observed using a FEI Tecnai F20 G2 transmission electron microscope (TEM). Images were acquired at an accelerating voltage of 50-200 kV under a nominal image resolution of < 0.23 nm, with magnifications ranging from ×25 to ×1,030,000.
2.7. Statistical analysis
All experiments were performed in triplicate (n = 3), and results are presented as mean ± standard deviation. Statistical analyses were conducted in R (version 4.3.3) using RStudio. Differences among treatments were evaluated by one-way analysis of variance (ANOVA) followed by Duncan’s multiple range test (p < 0.05). Pairwise comparisons were performed using Student’s t-test.
3. Results and Discussion
3.1. Effects of pre-treatment on SDF and IDF yields
Treatment significantly altered the partitioning of dietary fiber into soluble and insoluble fractions (Table 1). The control showed a low SDF yield (1.51%), whereas A. luchuensis 74-5 pre-treatment and the HCl-assisted process increased SDF yield to 10.17% and 12.00%, respectively (p < 0.05). Notably, the 74-5 workflow retained a higher IDF yield than the HCl condition (50.47% vs. 30.95%; p < 0.05). These trends are consistent with reports that fermentation-based pre-treatment can promote partial solubilization of fiber-rich matrices via microbial enzyme activities, thereby increasing extractable soluble fractions (Wang et al., 2024b: Al Faruq et al., 2025). Nevertheless, fermentation effects on soluble cell–wall polysaccharides are strain- and process-dependent, and changes in SDF may reflect a balance between matrix loosening and solubilization and enzymatic depolymerization (Tuly and Ma, 2024; Zhao et al., 2025; Meng et al., 2025). In one study, Aspergillus oryzae solid-state fermentation of apple peel was reported to decrease SDF while increasing IDF, suggesting that enzymatic depolymerization may offset solubilization under certain conditions (Li et al., 2024). Accordingly, the increased SDF observed in the present 74-5 workflow suggests that matrix loosening and solubilization predominated over enzymatic depolymerization under our fractionation conditions.
3.2. Hydration and dispersion properties of SDF and IDF fractions
In the SDF fraction, the control showed low solubility (47.78%) and high water absorption (2.89 g/g), and its dispersibility was not measurable (NM). By contrast, the 74-5 workflow produced a markedly more water-soluble fraction (93.19%) with substantially lower water absorption (0.80 g/g) than the HCl-derived SDF (49.39% and 2.60 g/g) (Table 1; p < 0.05), while showing comparable dispersibility (43.23% vs. 45.72%). In the IDF fraction, the control exhibited the highest apparent solubility (30.34%), water absorption (5.39 g/g), and dispersibility (90.27%), whereas HCl-IDF showed the lowest apparent solubility (8.96%) and dispersibility (7.22%) (Table 1). Compared with HCl-IDF, 74-5 IDF retained higher apparent solubility (15.17%) and dispersibility (15.22%) with similar water absorption (3.90 vs. 3.70 g/g). Overall, relative to the control, both pre-treatments shifted fiber partitioning and hydration behavior, with the 74-5 workflow yielding a more soluble SDF while preserving a larger IDF fraction than the HCl-assisted process. Collectively, these results suggest that solubility, water absorption, and dispersibility do not necessarily covary in apple pomace-derived SDF and IDF fractions: process-driven cell-wall disruption may increase the solubilized fraction without increasing water uptake, and dispersibility may also depend on particle wettability and microstructure, as reported in previous studies (Schmid et al., 2020; Schmid et al., 2021; Hollestelle et al., 2024). Accordingly, these findings suggest that hydration and dispersion metrics may be process–dependent, such that solubility, water uptake, and dispersibility do not necessarily show a fixed positive association.
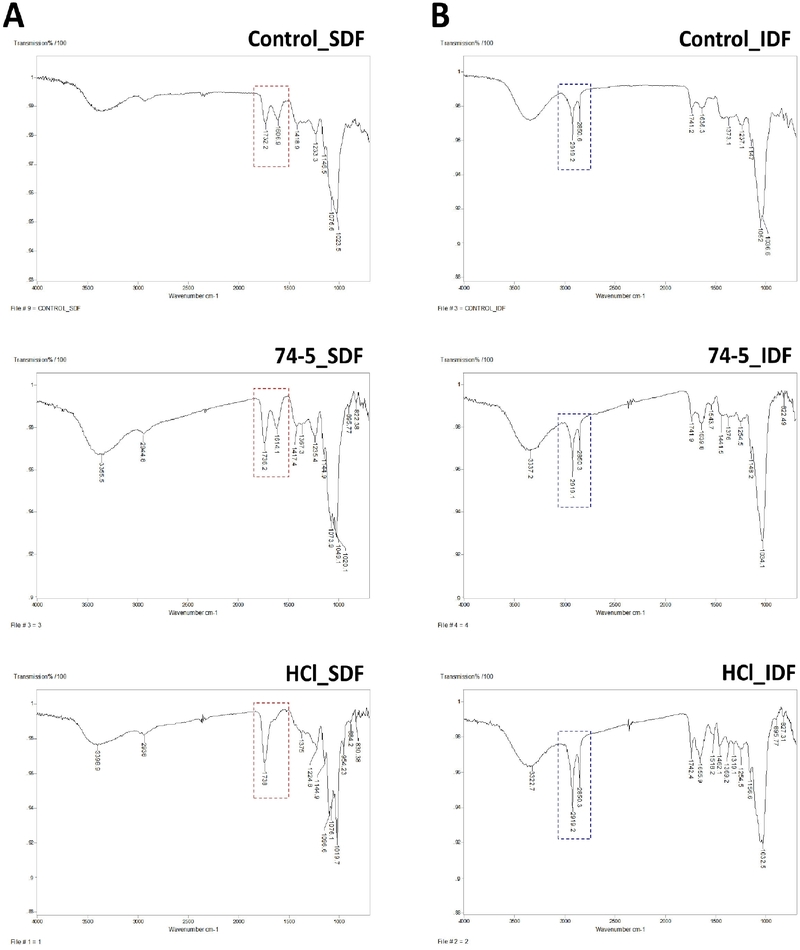
3.3. FT-IR spectra of SDF and IDF fractions
In the SDF spectra (Fig. 2A, red-highlighted), bands at 1730-1745 cm⁻¹ and 1600-1630 cm⁻¹ were observed across treatments and are attributable to pectic ester carbonyl (C=O) and carboxylate (COO⁻) groups, respectively (Jarrín-Chacón et al., 2023; Santiago-Gómez et al., 2025). Because the relative intensity of these bands is commonly associated with the methyl-esterification state of pectic carboxyl groups, the observed profiles suggest pre–treatment-dependent differences in pectic functional–group signatures. Notably, the HCl–derived SDF showed a relatively more pronounced band near 1730-1745 cm⁻¹ than the control and 74-5 SDFs, consistent with an acid-associated shift in the balance between ester carbonyl and carboxylate contributions under the present conditions. By contrast, the IDF spectra (Fig. 2B, blue-highlighted) were dominated by the aliphatic C-H stretching region at 2950-2850 cm⁻¹, with distinct bands near 2920 and 2890 cm⁻¹ across treatments, consistent with plant cell-wall materials (e.g., cellulose-rich fibers) (Meraj et al., 2024; Kaya, 2024).
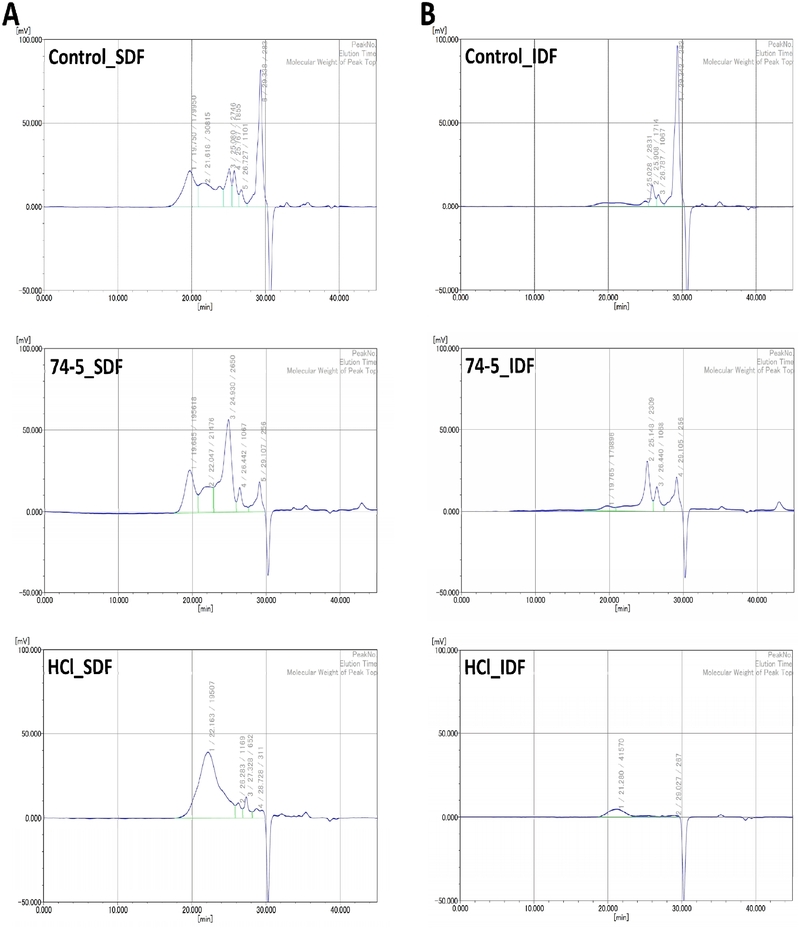
3.4. GPC chromatograms of SDF and IDF fractions
GPC chromatograms showed that pre-treatment altered polymer populations in both SDF and IDF fractions by shifting the molecular-weight distribution (MWD) (Fig. 3). These changes were reflected in differences in dominant peak-top molecular weights (Mp) and dispersity (Mw/Mn) (PEG/PEO-equivalent; RI-based). For the SDF fraction (Fig. 3A), Control_SDF exhibited a broad, multi-peaked distribution (Mw/Mn = 114). The 74–5 pre-treatment shifted the distribution toward a dominant low-kDa population while retaining a high-MW tail (Mw/Mn = 33.4), consistent with reports that fermentation can remodel pectin populations during subsequent aqueous extraction (Xu et al., 2022; Muslu Can et al., 2024). In contrast, HCl_SDF showed the narrowest distribution centered around a mid-kDa population (Mw/Mn = 12.7), consistent with acid-catalyzed depolymerization and peak “focusing” under heated acidic extraction (Ma et al., 2025b; Pattarapisitporn and Noma, 2025). For the IDF fraction (Fig. 3B), Control_IDF was dominated by low-kDa components with high dispersity (Mw/Mn = 70.7). The 74-5 pre-treatment increased the relative contribution of a low-kDa population while retaining a minor high-MW tail (Mw/Mn = 56.5), whereas HCl_IDF displayed a simplified two-population profile with reduced dispersity (Mw/Mn = 25.4), consistent with acid hydrolysis-driven redistribution of polymer size classes (Ma et al., 2025b; Pattarapisitporn and Noma, 2025).
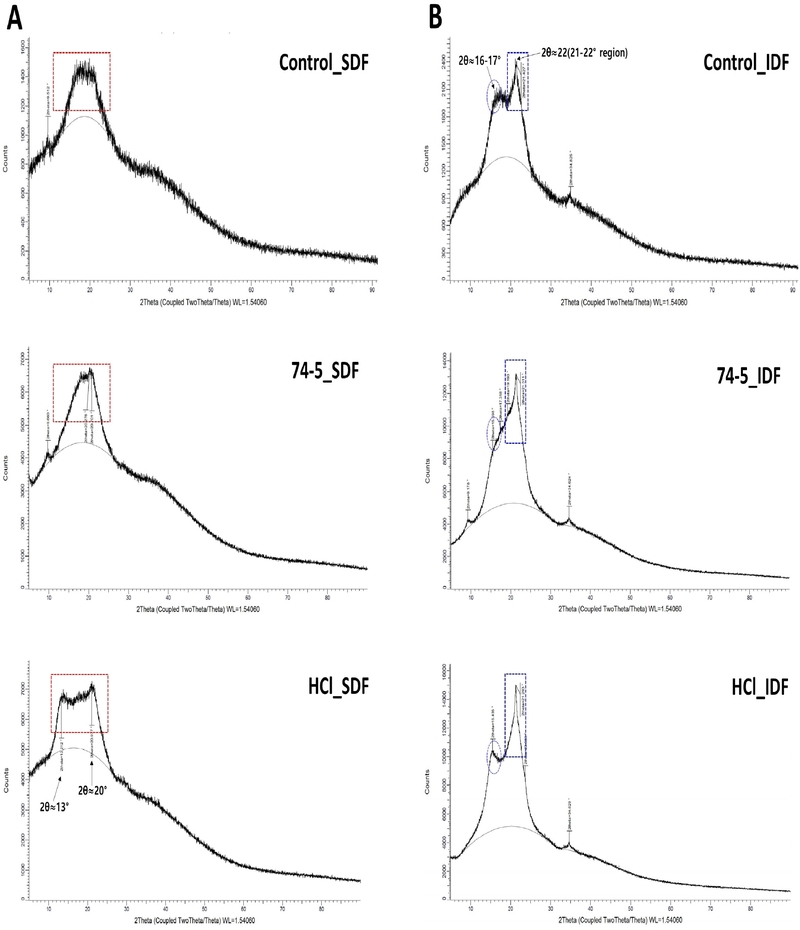
3.5. XRD patterns of SDF and IDF fractions
In the SDF patterns (Fig. 4A), all treatments showed a broad halo in the 13-22° (2θ) region (red boxes) with no prominent crystalline reflections, indicating predominantly amorphous polysaccharide matrices (Sayed et al., 2022). By contrast, the IDF patterns (Fig. 4B) showed cellulose-associated diffraction peaks, including a strong peak at 2θ≈22° (blue boxes) and a weak shoulder feature near 16-17° (blue circles), indicating crystalline cellulose domains in the insoluble fraction.
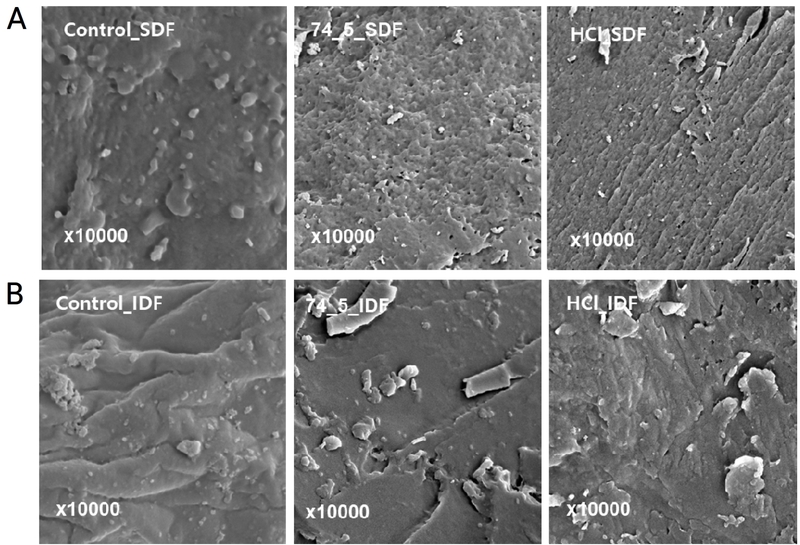
3.6. Microstructure and morphology assessed by SEM and TEM
In the SDF SEM images (Fig. 5A), 74-5_SDF exhibited a rough, microvoid-rich surface compared with the more compact, sheet-like appearance of Control_SDF and HCl_SDF, consistent with reports that processing can alter the porosity and surface texture of dietary-fiber/pectin-rich materials (Wu et al., 2022; Qin et al., 2024). In the IDF SEM images (Fig. 5B), all treatments largely retained a plate-like morphology, although surface roughness and particulate deposits differed among treatments. Overall, the more open surface texture of 74-5_SDF could facilitate wetting and water penetration, qualitatively consistent with its higher solubility and lower water absorption in Table 1.
In the SDF TEM images (Fig. 6A), Control_SDF showed a largely uniform background with only occasional electron-dense particulates. By contrast, 74-5_SDF exhibited more frequent, finely dispersed electron-dense nanoscale particulates, whereas HCl_SDF showed fewer but larger electron-dense clusters. Together, these patterns suggest that, even with an identical ethanol-precipitation step, upstream pre–treatment and extraction conditions may influence the nanoscale association of the recovered SDF, with 74-5 pre-treatment tending toward finer dispersion and the HCl-assisted route tending toward larger clusters (Guo et al., 2017; Chen et al., 2021). In the IDF TEM images (Fig. 6B), Control_IDF displayed a compact electron-dense aggregate, while 74-5_IDF showed smaller and more dispersed particulates across the field. In contrast, HCl_IDF exhibited an extensive, interconnected web-like fibrillar network. Such network-like nanofibrillar morphologies have been reported for fibrous substrates after chemical or enzyme-assisted processing, supporting the interpretation that pre-treatment altered the nanoscale organization of the insoluble matrix (Chen et al., 2015; Xie et al., 2016).
3.7. Environmental implications of workflow selection
Compared with conventional acid-assisted extraction, the fermentation-based workflow reduces reliance on strong acids, thereby potentially decreasing the generation of acidic process streams and the associated needs for neutralization and wastewater treatment (Bosch and Malgas, 2023; Dang et al., 2025). This shift is environmentally relevant because life-cycle and process sustainability assessments of acid–based extraction systems consistently indicate that reagent consumption and downstream conditioning steps can be major contributors to overall burdens, meaning that chemical-leaner routes can translate into measurable reductions in impact (Midolo et al., 2024; Carpentieri et al., 2025; del Amo-Mateos et al., 2026). In particular, since wastewater treatment is typically energy-intensive and closely linked to greenhouse-gas emissions, lowering neutralization demand and treatment load could reduce the indirect energy use and carbon footprint of the overall fractionation workflow (Ramírez-Melgarejo and Stringer, 2024; El Abbadi et al., 2025; Lan Anh and Le Luu, 2025).
Diverting AP from landfilling to valorization workflows is environmentally important because methane is generated when biodegradable organics decompose under anaerobic landfill conditions. To mitigate landfill methane, management measures generally combine reduced biodegradable inputs with earlier landfill-gas capture for destruction or energy recovery (Scharff et al., 2024; Dai and Wang, 2024; Folorunsho et al., 2026). In this framework, diverting moisture-rich AP into SDF and IDF fractionation represents a practical form of input reduction and may therefore lower methane-related impacts relative to anaerobic landfill disposal (Chickering et al., 2023; Scharff et al., 2024; Kaban et al., 2025). In this context, a lower-chemical fermentation route may deliver more specific climate benefits by (i) avoiding fugitive CH₄ by diverting wet AP away from landfilling and (ii) lowering indirect emissions linked to strong-acid use through reduced neutralization demand and wastewater-treatment burden. Future studies should evaluate the net climate benefit of the fermentation-based route using a comparative life cycle assessment (LCA) following standard LCA frameworks. This assessment should account for process energy use, chemical inputs, downstream neutralization and wastewater-treatment requirements, and avoided emissions relative to landfilling.
4. Conclusions
This study compared a fermentation–based pre-treatment (A. luchuensis 74–5) with an HCl-assisted route for fractionating apple pomace into SDF and IDF. The 74-5 workflow increased SDF yield from 1.51% to 10.17%, approaching the HCl-assisted process (12.00%), while retaining a higher IDF yield than HCl (50.47% vs. 30.95%; p < 0.05). The 74-5-derived SDF exhibited markedly higher solubility (93.19%) and lower water absorption (0.80 g/g) than the HCl-derived SDF (49.39% and 2.60 g/g), with comparable dispersibility. FT-IR, GPC, XRD, SEM, and TEM analyses supported workflow-dependent differences in structural signatures and microstructure across treatments.
Overall, the 74-5-based workflow (fermentation pre-treatment-hot-water extraction-ethanol precipitation) represents a reduced-chemical option for producing functionality-oriented SDF and IDF fractions from apple pomace. By avoiding strong–acid extraction, this workflow may reduce chemical inputs as well as downstream neutralization and wastewater-treatment requirements. Moreover, diverting apple pomace from anaerobic disposal pathways such as landfilling may offer potential to mitigate methane-related emissions and the associated energy and carbon burdens compared with acid-based processing.
Acknowledgments
This research was funded by the Research Program for Agriculture Science and Technology Development (Project No. PJ01733201), Rural Development Administration (RDA), Republic of Korea, and by the 2024–2026 RDA Postdoctoral Fellowship Program of the National Institute of Crop and Food Science (NICS), RDA.
REFERENCES
-
Al Faruq, A., Farahnaky, A., Torley, P. J., Buckow, R., Eri, R., Majzoobi, M., 2025, Sustainable approaches to boost soluble dietary fibre in foods: A Path to healthier foods, Food Hydrocolloids, 162, 110880.
[https://doi.org/10.1016/j.foodhyd.2024.110880]

-
Ampese, L. C., Ziero, H. D. D., Velásquez, J., Sganzerla, W. G., Martins, G., Forster-Carneiro, T., 2023, Apple pomace management by anaerobic digestion and composting: A Life cycle assessment, Biofuels, Bioprod. Biorefin., 17(1), 29-45.
[https://doi.org/10.1002/bbb.2446]

-
Antonic, B., Jancikova, S., Dordevic, D., Tremlova, B., 2020, Apple pomace as food fortification ingredient: A Systematic review and meta-analysis, J. Food Sci., 85(10), 2977-2985.
[https://doi.org/10.1111/1750-3841.15449]

-
Asif, M., Javaid, T., Razzaq, Z. U., Khan, M. K. I., Maan, A. A., Yousaf, S., Usman, A., Shahid, S., 2024, Sustainable utilization of apple pomace and its emerging potential for development of functional foods, Environ. Sci. Pollut. Res. Int., 31(12), 17932-17950.
[https://doi.org/10.1007/s11356-023-28479-9]

-
Azarakhsh, F. A. S., Ziloue, H., Ebrahimian, F., Khoshnevisan, B., Denayer, J. F. M., Karimi, K., 2024, Life cycle analysis of apple pomace biorefining for biofuel and pectin production, Sci. Total Environ., 951, 175780.
[https://doi.org/10.1016/j.scitotenv.2024.175780]

-
Ben-Othman, S., Jõudu, I., Bhat, R., 2020, Bioactives from agri-food wastes: Present insights and future challenges, Molecules, 25(3), 510.
[https://doi.org/10.3390/molecules25030510]

-
Bosch, R., Malgas, S., 2023, Ultrasound-assisted enzymatic extraction of orange peel pectin and its characterisation, Int. J. Food Sci. Technol., 58, 6784-6793.
[https://doi.org/10.1111/ijfs.16646]

-
Buljeta, I., Šubarić, D., Babić, J., Pichler, A., Šimunović, J., Kopjar, M., 2023, Extraction of dietary fibers from plant-based industry waste: a comprehensive review, Appl. Sci., 13(16), 9309.
[https://doi.org/10.3390/app13169309]

-
Carpentieri, S., Ghanem, A., Khwaldia, K., Silva, A. S., Ferrari, G., 2025, Life cycle assessment of agro-industrial residues valorization processes to obtain phenolic-rich extracts, Front. Sustain. Food Syst., 9, 1693181.
[https://doi.org/10.3389/fsufs.2025.1693181]

-
Chaudhary, N., Tiwari, V., Sharma, A., Kumari, A., Garg, M., Bhatnagar, A., Kansal, S. K., Krishania, M., 2024, New strategy for browning prevention in apple pomace processing and toxicity tested in a rodent model, ACS Omega, 9(30), 33022-33032.
[https://doi.org/10.1021/acsomega.4c03966]

-
Chen, R., Ratcliffe, I., Williams, P. A., Luo, S., Chen, J., Liu, C., 2021, The influence of pH and monovalent ions on the gelation of pectin from the fruit seeds of the creeping fig plant, Food Hydrocolloids, 111, 106219.
[https://doi.org/10.1016/j.foodhyd.2020.106219]

-
Chen, W., Li, Q., Cao, J., Liu, Y., Li, J., Zhang, J., Luo, S., Yu, H., 2015, Revealing the structures of cellulose nanofiber bundles obtained by mechanical nanofibrillation via TEM observation, Carbohydr. Polym., 117, 950-956.
[https://doi.org/10.1016/j.carbpol.2014.10.024]

-
Chickering, G., Krause, M. J., Schwarber, A., 2023, Effects of landfill food waste diversion: A Focus on microbial populations and methane generation, Biodegradation, 34(5), 477-488.
[https://doi.org/10.1007/s10532-023-10034-5]

-
Costa, J. M., Ampese, L. C., Ziero, H. D. D., Sganzerla, W. G., Forster-Carneiro, T., 2022, Apple pomace biorefinery: Integrated approaches for the production of bioenergy, biochemicals, and value–added products – An Updated review, J. Environ. Chem. Eng., 10(5), 108358.
[https://doi.org/10.1016/j.jece.2022.108358]

-
Czubaszek, R., Wysocka-Czubaszek, A., Tyborowski, R., 2022, Methane production potential from apple pomace, cabbage leaves, pumpkin residue and walnut husks, Appl. Sci., 12(12), 6128.
[https://doi.org/10.3390/app12126128]

-
Dai, F., Wang, Y., 2024, Mitigating methane emissions: Domestic and joint efforts by the United States and China, Environ. Sci. Ecotechnology, 20, 100398.
[https://doi.org/10.1016/j.ese.2024.100398]

-
Dang, G., Li, J., Yin, C., Wang, W., Zhang, K., Zhong, R., Chen, L., Zhang, H., Schroyen, M., 2025, Deciphering pectin: A Comprehensive overview of its origins, processing, and promising utility, ACS Omega, 10, 1-15.
[https://doi.org/10.1021/acsomega.4c01843]

-
del Amo-Mateos, E., Gosalvitr, P., Cuéllar-Franca, R. M., Dragone, G., Mussatto, S. I., García-Cubero, M. T., Coca, M., Lucas, S., 2026, An Environmental and economic sustainability assessment of novel rhamnogalacturonan-I pectin production from agricultural residues, J. Environ. Chem. Eng., 14, 120786.
[https://doi.org/10.1016/j.jece.2025.120786]

-
El Abbadi, S. H., Feng, J., Hodson, A. R., Amouamouha, M., Busse, M., Polcuch, C., Zhou, P., Macknick, J., Guest, J. S., Stokes-Draut, J. R., Dunn, J. B., 2025, Benchmarking greenhouse gas emissions from US wastewater treatment for targeted reduction, Nat. Water, 3, 1133-1143.
[https://doi.org/10.1038/s44221-025-00485-w]

-
Folorunsho, A., Dudhia, J., Jiang, L., Yokochi, A., Li, Y., 2026, Assessing climate and air quality benefits of methane mitigation for U.S. landfills, J. Environ. Manage., 400, 128707.
[https://doi.org/10.1016/j.jenvman.2026.128707]

-
Futagami, T., 2022, The white koji fungus Aspergillus luchuensis mut. kawachii, Biosci. Biotechnol. Biochem., 86(5), 574-584.
[https://doi.org/10.1093/bbb/zbac033]

-
Guo, X., Zhang, T., Meng, H., Yu, S., 2017, Ethanol precipitation of sugar beet pectins as affected by electrostatic interactions between counter ions and pectin chains, Food Hydrocolloids, 65, 187-197.
[https://doi.org/10.1016/j.foodhyd.2016.11.010]

-
Ishara, G. K. K., Koliyabandara, P. A., Samarakoon, G., 2024, Eco–friendly bio–composite sheets: A Study on the utilization of banana peels, cassava starch, and banana stem fibers, Front. Sustain., 5, 1410986.
[https://doi.org/10.3389/frsus.2024.1410986]

-
Jarrín-Chacón, J. P., Núñez-Pérez, J., Espín-Valladares, R. D. C., Manosalvas-Quiroz, L. A., Rodríguez-Cabrera, H. M., Pais-Chanfrau, J. M., 2023, Pectin extraction from residues of the cocoa fruit (Theobroma cacao L.) by different organic acids: A Comparative study, Foods, 12(3), 590.
[https://doi.org/10.3390/foods12030590]

-
Jinapong, N., Suphantharika, M., Jamnong, P., 2008, Production of instant soymilk powders by ultrafiltration, spray drying and fluidized bed agglomeration, J. Food Eng., 84(2), 194-205.
[https://doi.org/10.1016/j.jfoodeng.2007.04.032]

-
Kaban, G., Gürakar, B., Sayın, B., Polat, Z., Kaya, M., 2025, Utilization of apple pomace as a novel substrate for citric acid production by Yarrowia lipolytica, Sci. Rep., 15, 32890.
[https://doi.org/10.1038/s41598-025-99202-6]

-
Kaur, S., Singh, V., Panesar, P. S., Chopra, H. K., 2025, Microwave–assisted extraction of dietary fiber from kinnow mandarin by–products: Impact on yield and functional, structural, and thermal properties, Sustainable Food Technol., 3, 2122-2133.
[https://doi.org/10.1039/D5FB00362H]

-
Kaya, M., 2024, Characterization of TEMPO–oxidized cellulose nanofiber from biowaste and its influence on molecular behavior of fluorescent rhodamine B dye in aqueous suspensions, J. Fluoresc., 35(6), 4053-4063.
[https://doi.org/10.1007/s10895-024-03824-4]

- Korea Agro-Fisheries & Food Trade Corporation (aT), 2023, Fruit trends and statistics in South Korea, K–FoodTrade newsletter, Republic of Korea.
-
Kumari, T., Babu, S., Bag, K. K., Das, A. B., Deka, S. C., 2025, Dietary fiber modification: A Comparative study of physical, chemical, biological, combined technologies and bioactive impact on food applications, Carbohydr. Res., 554, 109558.
[https://doi.org/10.1016/j.carres.2025.109558]

-
Lan Anh, L. T., Le Luu, T., 2025, Greenhouse gas emissions from municipal wastewater treatment: Global insights and Vietnam's approach, Curr. Opin. Environ. Sci. Health, 47, 100655.
[https://doi.org/10.1016/j.coesh.2025.100655]

-
Li, R., Wang, T., Bo, N., Wang, Q., Chen, Q., Liang, Z., Guan, Y., Jiang, B., Ma, Y., Zhao, M., 2024, The carbohydrate metabolism and expression of carbohydrate-active enzyme genes in Aspergillus luchuensis fermentation of tea leaves, Front. Microbiol., 15, 1408645.
[https://doi.org/10.3389/fmicb.2024.1408645]

-
Liu, C.-M., Liang, L., Shuai, X.-X., Liang, R.-H., Chen, J., 2018, Dynamic high–pressure microfluidization–treated pectin under different ethanol concentrations, Polymers, 10(12), 1410.
[https://doi.org/10.3390/polym10121410]

-
Liu, T., Lei, H., Zhen, X., Liu, J., Xie, W., Tang, Q., Gou, D., Zhao, J., 2024, Advancements in modifying insoluble dietary fiber: Exploring the microstructure, physicochemical properties, biological activity, and applications in food industry—A Review, Food Chem., 458, 140154.
[https://doi.org/10.1016/j.foodchem.2024.140154]

-
Lyu, F., Luiz, S. F., Azeredo, D. R. P., Cruz, A. G., Ajlouni, S., Ranadheera, C. S., 2020, Apple pomace as a functional and healthy ingredient in food products: A Review, Processes, 8(3), 319.
[https://doi.org/10.3390/pr8030319]

-
Ma, M., Cui, J., Zhang, Y., Blecker, C., Zheng, J., 2025b, Changes in molecular structure of citrus pectin in acidic environments at ambient temperature, Carbohydr. Polym., 356, 123411.
[https://doi.org/10.1016/j.carbpol.2025.123411]

-
Ma, Y., Zheng, N., Wang, Y., Lei, H., Zhen, X., Zhang, R., Liu, T., 2025a, Insoluble dietary fiber stabilized Pickering emulsions as novel food ingredients: preparation, potential applications and future perspectives, Food Chem.: X, 27, 102458.
[https://doi.org/10.1016/j.fochx.2025.102458]

-
Mammolenti, D., Lupi, F. R., Baldino, N., Gabriele, D., 2025, Technological advancements of insoluble dietary fiber from food by–product processing: A Review, Foods, 14(10), 1822.
[https://doi.org/10.3390/foods14101822]

-
Meng, W., Hu, M., Zhang, P., Wang, J., Yuan, Z., Wang, F., Li, S., 2025, Efficient conversion of insoluble dietary fiber to soluble dietary fiber by Bacillus subtilis BSNK–5 fermentation of okara and improvement of their structural and functional properties, Food Chem., 474, 143188.
[https://doi.org/10.1016/j.foodchem.2025.143188]

-
Meraj, A., Jawaid, M., Singh, S. P., Nasef, M. M., Ariffin, H., Fouad, H., Abu-Jdayil, B., 2024, Isolation and characterisation of lignin using natural deep eutectic solvents pretreated kenaf fibre biomass, Sci. Rep., 14, 8672.
[https://doi.org/10.1038/s41598-024-59200-6]

-
Midolo, G., Cutuli, G., Porto, S. M. C., Ottolina, G., Paini, J., Valenti, F., 2024, LCA analysis for assessing environmenstal sustainability of new biobased chemicals by valorising citrus waste, Sci. Rep., 14, 21418.
[https://doi.org/10.1038/s41598-024-72468-y]

-
Muslu Can, A., Metin Yildirim, R., Karadag, A., 2024, The properties of pectin extracted from the residues of vinegar–fermented apple and apple pomace, Fermentation, 10(11), 556.
[https://doi.org/10.3390/fermentation10110556]

-
Pascoalino, L. A., Barros, L., Barreira, J. C. M., Oliveira, M. B. P. P., Reis, F. S., 2025, Closing the loop: Exploring apple pomace as a source of bioactive compounds in the framework of circular economy, Sustainable Food Technol., 3, 81-95.
[https://doi.org/10.1039/D4FB00172A]

-
Pattarapisitporn, A., Noma, S., 2025, Alternative solvents for pectin extraction: Effects of extraction agents on pectin structural characteristics and functional properties, Foods, 14(15), 2644.
[https://doi.org/10.3390/foods14152644]

-
Podgorbunskikh, E., Kuskov, T., Bukhtoyarov, V., Lomovsky, O., Bychkov, A., 2024, Recrystallization of cellulose, chitin and starch in their individual and native forms, Polymers, 16(7), 980.
[https://doi.org/10.3390/polym16070980]

-
Putra, N. R., Rizkiyah, D. N., Abdul Aziz, A. H., Che Yunus, M. A., Veza, I., Harny, I., Tirta, A., 2023, Waste to wealth of apple pomace valorization by past and current extraction processes: A Review, Sustainability, 15(1), 830.
[https://doi.org/10.3390/su15010830]

-
Qin, Y., Fan, X., Gao, Y., Wang, P., Chang, J., Liu, C., Wang, L., Yin, Q., 2024, Effects of physicochemical and biological treatment on structure, functional and prebiotic properties of dietary fiber from corn straw, Foods, 13(13), 1976.
[https://doi.org/10.3390/foods13131976]

-
Ramírez-Melgarejo, M., Stringer, T., 2024, Wastewater treatment, energy consumption, and greenhouse gas emissions: An Operational approach to comparing Barcelona and Mexico City, J. Environ. Manage., 353, 120175.
[https://doi.org/10.1016/j.jenvman.2024.120175]

-
Riyamol, Chengaiyan, J. G., Rana, S. S., Ahmad, F., Haque, S., Capanoglu, E., 2023, Recent advances in the extraction of pectin from various sources and industrial applications, ACS Omega, 8(49), 46309-46324.
[https://doi.org/10.1021/acsomega.3c04010]

-
Santiago-Gómez, I., Carrera-Lanestosa, A., González-Alejo, F. A., Guerra-Que, Z., García-Alamilla, R., Rivera-Armenta, J. L., García-Alamilla, P., 2025, Pectin extraction process from cocoa pod husk (Theobroma cacao L.) and characterization by Fourier transform infrared spectroscopy, ChemEngineering, 9(2), 25.
[https://doi.org/10.3390/chemengineering9020025]

-
Sayed, A., Hany, F., Abdel-Raouf, M. E.-S., Mahmoud, G. A., 2022, Gamma irradiation synthesis of pectin–based biohydrogels for removal of lead cations from simulated solutions, J. Polym. Res., 29, 372.
[https://doi.org/10.1007/s10965-022-03219-8]

-
Scharff, H., Soon, H.-Y., Taremwa, S. R., Zegers, D., Dick, B., Villas Bôas Zanon, T., Shamrock, J., 2024, The impact of landfill management approaches on methane emissions, Waste Manag. Res., 42(11), 1052-1064.
[https://doi.org/10.1177/0734242X231200742]

-
Sifat, S. A. D., Trisha, A. T., Huda, N., Zzaman, W., Julmohammad, N., 2021, Response surface approach to optimize the conditions of foam mat drying of plum in relation to the physical–chemical and antioxidant properties of plum powder, Int. J. Food Sci., 2021, 3681807.
[https://doi.org/10.1155/2021/3681807]

-
Szymańska-Chargot, M., Chylińska, M., Gdula, K., Kozioł, A., Zdunek, A., 2017, Isolation and characterization of cellulose from different fruit and vegetable pomaces, Polymers, 9(10), 495.
[https://doi.org/10.3390/polym9100495]

-
Thomas, F., Abebe, G., Emenike, C., Martynenko, A., 2025, Sustainable utilization of apple pomace: Technological aspects and emerging applications, Food Res. Int., 220, 117149.
[https://doi.org/10.1016/j.foodres.2025.117149]

-
Tuly, J. A., Ma, H., 2024, Bioconversion of food industrial waste okara by microbial fermentation: Scope of omics study and possibility, Trends Food Sci. Technol., 146, 104391.
[https://doi.org/10.1016/j.tifs.2024.104391]

- USDA Foreign Agricultural Service (FAS), 2023, Korea’s apple update, GAIN Report, KS2023-0022, Seoul, Republic of Korea.
-
Vandorou, M., Plakidis, C., Tsompanidou, I. M., Adamantidi, T., Panagopoulou, E. A., Tsoupras, A., 2024, A review on apple pomace bioactives for natural functional food and cosmetic products with therapeutic health–promoting properties, Int. J. Mol. Sci., 25(19), 10856.
[https://doi.org/10.3390/ijms251910856]

-
Wang, G., Xie, L., Huang, Z., Xie, J., 2024a, Recent advances in polysaccharide biomodification by microbial fermentation: production, properties, bioactivities, and mechanisms, Crit. Rev. Food Sci. Nutr., 64, 12999-13023.
[https://doi.org/10.1080/10408398.2023.2259461]

-
Wang, H., Li, M., Jiao, F., Ge, W., Liu, R., Zhi, Z., Wu, T., Sui, W., Zhang, M., 2024b, Soluble dietary fibers from solid–state fermentation of wheat bran by the fungus Cordyceps cicadae and their effects on colitis mice, Food Funct., 15, 516-529.
[https://doi.org/10.1039/D3FO03851C]

-
Wu, Z., Qin, D., Li, H., Guo, D., Cheng, H., Sun, J., Huang, M., Ye, X., Sun, B., 2022, Physicochemical and functional properties of Lycium ruthenicum pectin by different extraction methods, Front. Nutr., 9, 946606.
[https://doi.org/10.3389/fnut.2022.946606]

-
Xie, J., Hse, C.-Y., De Hoop, C. F., Hua, T., Qi, J., Shupe, T. F., 2016, Isolation and characterization of cellulose nanofibers from bamboo using microwave liquefaction combined with chemical treatment and ultrasonication, Carbohydr. Polym., 151, 725-734.
[https://doi.org/10.1016/j.carbpol.2016.06.011]

-
Xu, F., Zhang, S., Waterhouse, G. I. N., Zhou, T., Du, Y., Sun-Waterhouse, D., Wu, P., 2022, Yeast fermentation of apple and grape pomaces affects subsequent aqueous pectin extraction: Composition, structure, functional and antioxidant properties of pectins, Food Hydrocolloids, 133, 107945.
[https://doi.org/10.1016/j.foodhyd.2022.107945]

-
Yamashita, H., 2021, Koji starter and koji world in Japan, J. Fungi., 7(7), 569.
[https://doi.org/10.3390/jof7070569]

-
Zaky, A. A., Witrowa-Rajchert, D., Nowacka, M., 2024, Turning apple pomace into value: Sustainable recycling in food production—A Narrative review, Sustainability, 16(16), 7001.
[https://doi.org/10.3390/su16167001]

-
Zhao, Y., Shao, Y., Fan, S., Bai, J., Zhu, L., Zhu, Y., Xiao, X., 2025, Advanced modification strategies of plant–sourced dietary fibers and their applications in functional foods, Foods, 14(15), 2710.
[https://doi.org/10.3390/foods14152710]

Food Tech Resources Research Division, National Institute of Crop and Food Science (NICS), Rural Development Administration (RDA)E-mail:hjwon7943@korea.kr
Food Tech Resources Research Division, National Institute of Crop and Food Science (NICS), Rural Development Administration (RDA)E-mail:dahee215@korea.kr
Food Tech Resources Research Division, National Institute of Crop and Food Science (NICS), Rural Development Administration (RDA)E-mail:yanghs0704@korea.kr
Food Tech Resources Research Division, National Institute of Crop and Food Science (NICS), Rural Development Administration (RDA)E-mail:kyk314@korea.kr
Food Tech Resources Research Division, National Institute of Crop and Food Science (NICS), Rural Development Administration (RDA)E-mail:lshin@korea.kr
Food Tech Resources Research Division, National Institute of Crop and Food Science (NICS), Rural Development Administration (RDA)E-mail:s2y337@korea.kr
Food Tech Resources Research Division, National Institute of Crop and Food Science (NICS), Rural Development Administration (RDA)E-mail:kmyqwer@korea.kr
Food Tech Resources Research Division, National Institute of Crop and Food Science (NICS), Rural Development Administration (RDA)E-mail:kimmina@korea.kr
Food Tech Resources Research Division, National Institute of Crop and Food Science (NICS), Rural Development Administration (RDA)E-mail:aejini77@korea.kr